Efficacy and safety of gemcitabine plus S-1 vs. gemcitabine plus nab-paclitaxel in treatment-naïve advanced pancreatic ductal adenocarcinoma
- PMID: 37646237
- PMCID: PMC10618946
- DOI: 10.20892/j.issn.2095-3941.2023.0189
Efficacy and safety of gemcitabine plus S-1 vs. gemcitabine plus nab-paclitaxel in treatment-naïve advanced pancreatic ductal adenocarcinoma
Abstract
Objective: Gemcitabine plus nab-paclitaxel (GnP) is the standard first-line therapy for advanced pancreatic ductal adenocarcinoma (PDAC). S-1, an oral fluoropyrimidine derivative, as compared with gemcitabine, is non-inferior in terms of overall survival (OS) and is associated with lower hematologic toxicity. Accordingly, S-1 is a convenient oral alternative treatment for advanced PDAC. This study was aimed at comparing the efficacy and safety of gemcitabine plus S-1 (GS) vs. GnP as first-line chemotherapy for advanced PDAC.
Methods: Patients with advanced PDAC who received first-line GS or GnP at the Peking Union Medical College Hospital between March 2011 and November 2022 were evaluated.
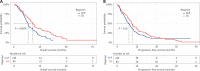
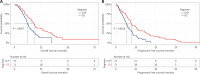
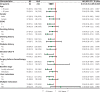
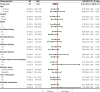
Results: A total of 300 patients were assessed, of whom 84 received GS and 216 received GnP. The chemotherapy completion rate was higher with GS than GnP (50.0% vs. 30.3%, P = 0.0028). The objective response rate (ORR) was slightly higher (14.3% vs. 9.7%, P = 0.35), and the median OS was significantly longer (17.9 months vs. 13.3 months, P = 0.0078), in the GS group than the GnP group. However, the median progression-free survival (PFS) did not significantly differ between groups. Leukopenia risk was significantly lower in the GS group than the GnP group (14.9% vs. 28.1%, P = 0.049).
Conclusions: As first-line chemotherapy for advanced PDAC, the GS regimen led to a significantly longer OS than the GnP regimen. The PFS, ORR, and incidence of severe adverse events were comparable between the GS and GnP groups.
Keywords: Advanced pancreatic cancer; S-1; first-line chemotherapy; gemcitabine; nab-paclitaxel.
Copyright © 2023 Cancer Biology & Medicine.
Conflict of interest statement
No potential conflicts of interest are disclosed.
Figures
Similar articles
-
Effect of a MUC5AC Antibody (NPC-1C) Administered With Second-Line Gemcitabine and Nab-Paclitaxel on the Survival of Patients With Advanced Pancreatic Ductal Adenocarcinoma: A Randomized Clinical Trial.JAMA Netw Open. 2023 Jan 3;6(1):e2249720. doi: 10.1001/jamanetworkopen.2022.49720. JAMA Netw Open. 2023. PMID: 36602796 Free PMC article. Clinical Trial.
-
Efficacy and Safety of Nab-Paclitaxel Plus S-1 versus Nab-Paclitaxel Plus Gemcitabine for First-Line Chemotherapy in Advanced Pancreatic Ductal Adenocarcinoma.Cancer Manag Res. 2020 Dec 9;12:12657-12666. doi: 10.2147/CMAR.S263773. eCollection 2020. Cancer Manag Res. 2020. PMID: 33328763 Free PMC article.
-
A Phase I Study of Gemcitabine/Nab-Paclitaxel/S-1 Chemotherapy in Patients With Locally Advanced or Metastatic Pancreatic Ductal Adenocarcinoma.Oncologist. 2023 Jul 5;28(7):e575-e584. doi: 10.1093/oncolo/oyac146. Oncologist. 2023. PMID: 35930304 Free PMC article. Clinical Trial.
-
Efficacy and safety of neoadjuvant Folfirinox and Gemcitabine plus Nab-Paclitaxel for borderline resectable and locally advanced pancreatic cancer: a systematic review and meta-analysis.Eur Rev Med Pharmacol Sci. 2022 Sep;26(17):6316-6327. doi: 10.26355/eurrev_202209_29656. Eur Rev Med Pharmacol Sci. 2022. PMID: 36111933
-
Clinical outcome of patients with inoperable pancreatic cancer treated with FOLFIRINOX or gemcitabine plus Nab‑paclitaxel as a first‑line therapy: A retrospective analysis.Med Int (Lond). 2021 Jul 5;1(4):8. doi: 10.3892/mi.2021.8. eCollection 2021 Sep-Oct. Med Int (Lond). 2021. PMID: 38939364 Free PMC article. Review.
Cited by
-
Programmed cell death 1 inhibitor sintilimab plus S-1 and gemcitabine for liver metastatic pancreatic ductal adenocarcinoma.World J Clin Oncol. 2025 Feb 24;16(2):98079. doi: 10.5306/wjco.v16.i2.98079. World J Clin Oncol. 2025. PMID: 39995563 Free PMC article.
-
Clinical outcomes of second-line chemotherapy in patients with advanced pancreatic adenocarcinoma: a real-world study.Cancer Biol Med. 2024 Jul 26;21(9):799-812. doi: 10.20892/j.issn.2095-3941.2024.0036. Cancer Biol Med. 2024. PMID: 39066473 Free PMC article.
References
-
- Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74:2913–21. - PubMed
-
- Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48. - PubMed
-
- Burris HA, 3rd, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15:2403–13. - PubMed
-
- Conroy T, Desseigne F, Ychou M, Bouche O, Guimbaud R, Becouarn Y, et al. FOLFIRINOX vs. gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364:1817–25. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
