Conjugated linoleic acids inhibit lipid deposition in subcutaneous adipose tissue and alter lipid profiles in serum of pigs
- PMID: 37646838
- PMCID: PMC10629446
- DOI: 10.1093/jas/skad294
Conjugated linoleic acids inhibit lipid deposition in subcutaneous adipose tissue and alter lipid profiles in serum of pigs
Abstract
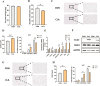
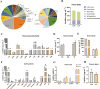
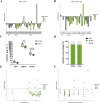
Conjugated linoleic acids (CLAs) have served as a nutritional strategy to reduce fat deposition in adipose tissues of pigs. However, the effects of CLAs on lipid profiles in serum and how these lipid molecules regulate fat deposition are still unclear. In this study, we explored the effects of CLAs on regulating lipid deposition in adipose tissues in terms of lipid molecules and microbiota based on a Heigai pig model. A total of 56 Heigai finishing pigs (body weight: 85.58 ± 10.39 kg) were randomly divided into two treatments and fed diets containing 1% soyabean oil or 1% CLAs for 40 d. CLAs reduced fat deposition and affected fatty acids composition in adipose tissues of Heigai pigs via upregulating the expression of the lipolytic gene (hormone-sensitive lipase, HSL) in vivo and in vitro. CLAs also altered the biochemical immune indexes including reduced content of total cholesterol (TChol), high-density lipoprotein (HDL-C), and low-density lipoprotein (LDL-C) and changed lipids profiles including decreased sphingolipids especially ceramides (Cers) and sphingomyelins (SMs) in serum of Heigai pigs. Mechanically, CLAs may decrease peroxisome proliferator-activated receptorγ (PPARγ) expression and further inhibit adipogenic differentiation in adipose tissues of pigs by suppressing the function of Cers in serum. Furthermore, Pearson's correlation analysis showed HSL expression was positively related to short-chain fatty acids (SCFAs) in the gut (P ≤ 0.05) but the abundance of Cers was negatively related to the production and functions of SCFAs (P ≤ 0.05). CLAs altered the distribution of the lipid in serum and inhibited adipogenic differentiation by suppressing the function of Cers and further decreasing PPARγ expression in adipose tissues of Heigai pigs. Besides, the HSL expression and the abundance of Cers are associated with the production and functions of SCFAs in the gut.
Keywords: ceramide; conjugated linoleic acids; fat deposition; lipidome; pig.
Plain language summary
Meat quality is affected by fat deposition and conjugated linoleic acids (CLAs) have served as a nutritional strategy to reduce fat deposition in adipose tissues of pigs. We explored the effects of CLAs on lipid profiles in serum and how these lipid molecules regulate fat deposition based on a Heigai pig model. We found CLAs reduced fat deposition in vivo and in vitro and changed lipids profiles in serum including decreased sphingolipids especially cermides (Cers) and sphingomyelins in the serum of Heigai pigs. We also demonstrated CLAs inhibited adipogenic differentiation by suppressing the function of Cers and further decreasing peroxisome proliferator-activated receptorγ expression in adipose tissues. Furthermore, Pearson’s correlation analysis showed hormone-sensitive lipase expression and the abundance of Cers are related to the production and functions of short-chain fatty acids in the gut. Our findings provide useful insights into the role of CLAs in regulating lipid composition in serum and lipid metabolism in adipose tissue and provide a new insight into producing high-quality pork in the pig industry by using nutritional strategies.
© The Author(s) 2023. Published by Oxford University Press on behalf of the American Society of Animal Science. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
The authors declare no conflict of interest, financial or otherwise.
Figures



Similar articles
-
CLA improves the lipo-nutritional quality of pork and regulates the gut microbiota in Heigai pigs.Food Funct. 2022 Nov 28;13(23):12093-12104. doi: 10.1039/d2fo02549c. Food Funct. 2022. PMID: 36377505
-
Changes in Serum Fatty Acid Composition and Metabolome-Microbiome Responses of Heigai Pigs Induced by Dietary N-6/n-3 Polyunsaturated Fatty Acid Ratio.Front Microbiol. 2022 Jun 22;13:917558. doi: 10.3389/fmicb.2022.917558. eCollection 2022. Front Microbiol. 2022. PMID: 35814644 Free PMC article.
-
Dietary conjugated linoleic acid differentially alters fatty acid composition and increases conjugated linoleic acid content in porcine adipose tissue.Br J Nutr. 2003 Nov;90(5):915-28. doi: 10.1079/bjn2003982. Br J Nutr. 2003. PMID: 14667185
-
Conjugated linoleic acids (CLAs) and white adipose tissue: how both in vitro and in vivo studies tell the story of a relationship.Histol Histopathol. 2006 Jun;21(6):663-72. doi: 10.14670/HH-21.663. Histol Histopathol. 2006. PMID: 16528676 Review.
-
[The impact of phenolic compounds on pig fat deposition].Sheng Wu Gong Cheng Xue Bao. 2024 May 25;40(5):1486-1497. doi: 10.13345/j.cjb.230498. Sheng Wu Gong Cheng Xue Bao. 2024. PMID: 38783810 Review. Chinese.
Cited by
-
Single-nucleus transcriptomics reveal the cytological mechanism of conjugated linoleic acids in regulating intramuscular fat deposition.Elife. 2025 Mar 7;13:RP99790. doi: 10.7554/eLife.99790. Elife. 2025. PMID: 40053468 Free PMC article.
-
Effects of dietary protein levels on genes related to subcutaneous fat deposition and lipid metabolism in Tibetan sheep.BMC Genomics. 2025 Jul 16;26(1):669. doi: 10.1186/s12864-025-11874-6. BMC Genomics. 2025. PMID: 40670965 Free PMC article.
References
-
- Blachnio-Zabielska, A. U., Baranowski M., Hirnle T., Zabielski P., Lewczuk A., Dmitruk I., and Gorski J... 2012. Increased bioactive lipids content in human subcutaneous and epicardial fat tissue correlates with insulin resistance. Lipids 47:1131–1141. doi: 10.1007/s11745-012-3722-x. - DOI - PMC - PubMed
-
- Chaurasia, B., Kaddai V. A., Lancaster G. I., Henstridge D. C., Sriram S., Galam D. L., Gopalan V., Prakash K. N., Velan S. S., Bulchand S.,. et al. 2016. Adipocyte ceramides regulate subcutaneous adipose browning, inflammation, and metabolism. Cell Metab. 24:820–834. doi: 10.1016/j.cmet.2016.10.002. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

