Profound Changes in Functional Structure and Dynamics of Serum Albumin in Children with Nephrotic Syndrome: An Exploratory Research Study
- PMID: 37648246
- PMCID: PMC10510392
- DOI: 10.1021/acs.jmedchem.3c00680
Profound Changes in Functional Structure and Dynamics of Serum Albumin in Children with Nephrotic Syndrome: An Exploratory Research Study
Abstract
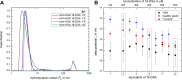
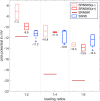
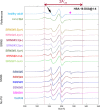
Patients with nephrotic syndrome (NS) suffer from urinary loss of albumin. As a cause, previous studies focused on the glomerular filter rather than analyzing the molecular properties of albumin itself. Later one was initiated by clinical observations indicating unexplained molecular alterations of human serum albumin (HSA) in an NS pediatric patient. Therefore, we examined serum from eight pediatric patients with steroid-sensitive and -resistant NS and compared it with serum from healthy subjects as well as commercial HSA. We used dynamic and electrophoretic light scattering to characterize the protein size and effective surface charge and electron paramagnetic resonance spectroscopy to measure the local environment and binding dynamics of up to seven fatty acids associated with HSA. Our findings suggest that pronounced differences in binding behavior and surface charge of HSA could enhance their filtration through the GBM, leading to direct toxicity of HSA to podocytes.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




Similar articles
-
Using bound fatty acids to disclose the functional structure of serum albumin.Biochim Biophys Acta. 2013 Dec;1830(12):5382-93. doi: 10.1016/j.bbagen.2013.04.031. Epub 2013 Apr 30. Biochim Biophys Acta. 2013. PMID: 23643928 Review.
-
Human Serum Albumin Loaded with Fatty Acids Reveals Complex Protein-Ligand Thermodynamics and Boleadora-Type Solution Dynamics Leading to Gelation.J Phys Chem B. 2025 Apr 10;129(14):3571-3589. doi: 10.1021/acs.jpcb.4c08717. Epub 2025 Mar 26. J Phys Chem B. 2025. PMID: 40138249 Free PMC article.
-
Circulating extracellular vesicles of patients with steroid-sensitive nephrotic syndrome have higher RAC1 and induce recapitulation of nephrotic syndrome phenotype in podocytes.Am J Physiol Renal Physiol. 2021 Nov 1;321(5):F659-F673. doi: 10.1152/ajprenal.00097.2021. Epub 2021 Sep 27. Am J Physiol Renal Physiol. 2021. PMID: 34569252 Free PMC article.
-
Binding of fatty acids facilitates oxidation of cysteine-34 and converts copper-albumin complexes from antioxidants to prooxidants.Arch Biochem Biophys. 2003 May 1;413(1):53-66. doi: 10.1016/s0003-9861(03)00091-2. Arch Biochem Biophys. 2003. PMID: 12706341
-
Fatty acid binding to serum albumin: molecular simulation approaches.Biochim Biophys Acta. 2013 Dec;1830(12):5427-34. doi: 10.1016/j.bbagen.2013.03.032. Epub 2013 Apr 6. Biochim Biophys Acta. 2013. PMID: 23567799 Review.
References
-
- Butt L.; Unnersjö-Jess D.; Höhne M.; Edwards A.; Binz-Lotter J.; Reilly D.; Hahnfeldt R.; Ziegler V.; Fremter K.; Rinschen M. M.; Helmstädter M.; Ebert L. K.; Castrop H.; Hackl M. J.; Walz G.; Brinkkoetter P. T.; Liebau M. C.; Tory K.; Hoyer P. F.; Beck B. B.; Brismar H.; Blom H.; Schermer B.; Benzing T. A Molecular Mechanism Explaining Albuminuria in Kidney Disease. Nat. Metab. 2020, 2, 461–474. 10.1038/s42255-020-0204-y. - DOI - PubMed
-
- Hausmann R.; Kuppe C.; Egger H.; Schweda F.; Knecht V.; Elger M.; Menzel S.; Somers D.; Braun G.; Fuss A.; Uhlig S.; Kriz W.; Tanner G.; Floege J.; Moeller M. J. Electrical Forces Determine Glomerular Permeability. J. Am. Soc. Nephrol. 2010, 21, 2053–2058. 10.1681/ASN.2010030303. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

