A short isoform of the UNC-6/Netrin receptor UNC-5 is required for growth cone polarity and robust growth cone protrusion in Caenorhabditis elegans
- PMID: 37649551
- PMCID: PMC10464613
- DOI: 10.3389/fcell.2023.1240994
A short isoform of the UNC-6/Netrin receptor UNC-5 is required for growth cone polarity and robust growth cone protrusion in Caenorhabditis elegans
Abstract
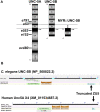
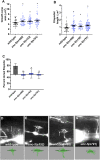
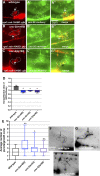
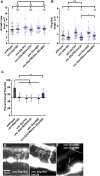
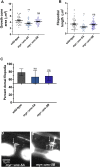
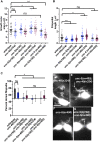
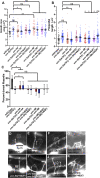
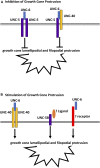
Introduction: UNC-6/Netrin is a conserved bi-functional guidance cue which regulates dorsal-ventral axon guidance in C. elegans. In the Polarity/Protrusion model of UNC-6/Netrin mediated dorsal growth away from UNC-6/Netrin, The UNC-5 receptor first polarizes the VD growth cone such that filopodial protrusions are biased dorsally. Based on this polarity, the UNC-40/DCC receptor stimulates growth cone lamellipodial and filopodial protrusion dorsally. The UNC-5 receptor maintains dorsal polarity of protrusion, and inhibits growth cone protrusion ventrally, resulting in net dorsal growth cone advance. Methods: Growth cone imaging in mutants, combined with Cas9 genome editing and genetic analysis, were used to analyze the role of a novel short isoform on unc-5 in growth cone polarity and protrusion. Results: Work presented here demonstrates a novel role of a previously undescribed, conserved short isoform of UNC-5 (UNC-5B). UNC-5B lacks the cytoplasmic domains of UNC-5 long, including the DEATH domain, the UPA/DB domain, and most of the ZU5 domain. Mutations that specifically affect only the unc-5 long isoforms were hypomorphic, suggesting a role of unc-5B short. A mutation specifically affecting unc-5B caused loss of dorsal polarity of protrusion and reduced growth cone filopodial protrusion, the opposite of unc-5 long mutations. Transgenic expression of unc-5B partially rescued unc-5 axon guidance defects, and resulted in large growth cones. Tyrosine 482 (Y482) in the cytoplasmic juxtamembrane region has been shown to be important for UNC-5 function, and is present in both UNC-5 long and UNC-5B short. Results reported here show that Y482 is required for the function of UNC-5 long and for some functions of UNC-5B short. Finally, genetic interactions with unc-40 and unc-6 suggest that UNC-5B short acts in parallel to UNC-6/Netrin to ensure robust growth cone lamellipodial protrusion. Discussion: These results demonstrate a previously-undescribed role for the UNC-5B short isoform, which is required for dorsal polarity of growth cone filopodial protrusion and to stimulate growth cone protrusion, in contrast to the previously-described role of UNC-5 long in inhibiting growth cone protrusion.
Keywords: UNC-6/Netrin; axon guidance; growth cone; polarity/protrusion; unc-5 netrin receptor B (UNC5B).
Copyright © 2023 Mahadik and Lundquist.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Update of
-
A short isoform of the UNC-6/Netrin receptor UNC-5 is required for growth cone polarity and robust growth cone protrusion in Caenorhabditis elegans.bioRxiv [Preprint]. 2023 May 3:2023.05.02.539117. doi: 10.1101/2023.05.02.539117. bioRxiv. 2023. Update in: Front Cell Dev Biol. 2023 Aug 15;11:1240994. doi: 10.3389/fcell.2023.1240994. PMID: 37205526 Free PMC article. Updated. Preprint.
Similar articles
-
Short- and long-range roles of UNC-6/Netrin in dorsal-ventral axon guidance in vivo in Caenorhabditis elegans.PLoS Genet. 2025 Jan 17;21(1):e1011526. doi: 10.1371/journal.pgen.1011526. eCollection 2025 Jan. PLoS Genet. 2025. PMID: 39823521 Free PMC article.
-
A short isoform of the UNC-6/Netrin receptor UNC-5 is required for growth cone polarity and robust growth cone protrusion in Caenorhabditis elegans.bioRxiv [Preprint]. 2023 May 3:2023.05.02.539117. doi: 10.1101/2023.05.02.539117. bioRxiv. 2023. Update in: Front Cell Dev Biol. 2023 Aug 15;11:1240994. doi: 10.3389/fcell.2023.1240994. PMID: 37205526 Free PMC article. Updated. Preprint.
-
Control of Growth Cone Polarity, Microtubule Accumulation, and Protrusion by UNC-6/Netrin and Its Receptors in Caenorhabditis elegans.Genetics. 2018 Sep;210(1):235-255. doi: 10.1534/genetics.118.301234. Epub 2018 Jul 25. Genetics. 2018. PMID: 30045855 Free PMC article.
-
Genetic analysis of growth cone migrations in Caenorhabditis elegans.J Neurobiol. 2000 Aug;44(2):281-8. J Neurobiol. 2000. PMID: 10934329 Review.
-
Netrin, Slit and Wnt receptors allow axons to choose the axis of migration.Dev Biol. 2008 Nov 15;323(2):143-51. doi: 10.1016/j.ydbio.2008.08.027. Epub 2008 Sep 5. Dev Biol. 2008. PMID: 18801355 Review.
Cited by
-
Short- and long-range roles of UNC-6/Netrin in dorsal-ventral axon guidance in vivo in Caenorhabditis elegans.PLoS Genet. 2025 Jan 17;21(1):e1011526. doi: 10.1371/journal.pgen.1011526. eCollection 2025 Jan. PLoS Genet. 2025. PMID: 39823521 Free PMC article.
-
SRC-1 controls growth cone polarity and protrusion with the UNC-6/Netrin receptor UNC-5 in Caenorhabditis elegans.PLoS One. 2024 May 21;19(5):e0295701. doi: 10.1371/journal.pone.0295701. eCollection 2024. PLoS One. 2024. PMID: 38771761 Free PMC article.
-
TRIM9 Controls Growth Cone Responses to Netrin Through DCC and UNC5C.J Neurochem. 2025 Jan;169(1):e70002. doi: 10.1111/jnc.70002. J Neurochem. 2025. PMID: 39871643
-
TIAM-1 regulates polarized protrusions during dorsal intercalation in the Caenorhabditis elegans embryo through both its GEF and N-terminal domains.J Cell Sci. 2024 Mar 1;137(5):jcs261509. doi: 10.1242/jcs.261509. Epub 2024 Mar 6. J Cell Sci. 2024. PMID: 38345070 Free PMC article.
-
Short- and long-range roles of UNC-6/Netrin in dorsal-ventral axon guidance in vivo in Caenorhabditis elegans.bioRxiv [Preprint]. 2024 Apr 23:2024.04.23.590737. doi: 10.1101/2024.04.23.590737. bioRxiv. 2024. Update in: PLoS Genet. 2025 Jan 17;21(1):e1011526. doi: 10.1371/journal.pgen.1011526. PMID: 38712249 Free PMC article. Updated. Preprint.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

