A two-step workflow based on plasma p-tau217 to screen for amyloid β positivity with further confirmatory testing only in uncertain cases
- PMID: 37653254
- PMCID: PMC10501903
- DOI: 10.1038/s43587-023-00471-5
A two-step workflow based on plasma p-tau217 to screen for amyloid β positivity with further confirmatory testing only in uncertain cases
Abstract
Cost-effective strategies for identifying amyloid-β (Aβ) positivity in patients with cognitive impairment are urgently needed with recent approvals of anti-Aβ immunotherapies for Alzheimer's disease (AD). Blood biomarkers can accurately detect AD pathology, but it is unclear whether their incorporation into a full diagnostic workflow can reduce the number of confirmatory cerebrospinal fluid (CSF) or positron emission tomography (PET) tests needed while accurately classifying patients. We evaluated a two-step workflow for determining Aβ-PET status in patients with mild cognitive impairment (MCI) from two independent memory clinic-based cohorts (n = 348). A blood-based model including plasma tau protein 217 (p-tau217), age and APOE ε4 status was developed in BioFINDER-1 (area under the curve (AUC) = 89.3%) and validated in BioFINDER-2 (AUC = 94.3%). In step 1, the blood-based model was used to stratify the patients into low, intermediate or high risk of Aβ-PET positivity. In step 2, we assumed referral only of intermediate-risk patients to CSF Aβ42/Aβ40 testing, whereas step 1 alone determined Aβ-status for low- and high-risk groups. Depending on whether lenient, moderate or stringent thresholds were used in step 1, the two-step workflow overall accuracy for detecting Aβ-PET status was 88.2%, 90.5% and 92.0%, respectively, while reducing the number of necessary CSF tests by 85.9%, 72.7% and 61.2%, respectively. In secondary analyses, an adapted version of the BioFINDER-1 model led to successful validation of the two-step workflow with a different plasma p-tau217 immunoassay in patients with cognitive impairment from the TRIAD cohort (n = 84). In conclusion, using a plasma p-tau217-based model for risk stratification of patients with MCI can substantially reduce the need for confirmatory testing while accurately classifying patients, offering a cost-effective strategy to detect AD in memory clinic settings.
© 2023. The Author(s).
Conflict of interest statement
O.H. has acquired research support (for the institution) from ADx, AVID Radiopharmaceuticals, Biogen, Eli Lilly, Eisai, Fujirebio, GE Healthcare, Pfizer and Roche. In the past 2 years, he has received consultancy/speaker fees from AC Immune, Amylyx, Alzpath, BioArctic, Biogen, Cerveau, Eisai, Eli Lilly, Fujirebio, Genentech, Merck, Novartis, Novo Nordisk, Roche, Sanofi and Siemens. K.B. has served as a consultant, at advisory boards, or at data monitoring committees, for Abcam, Axon, BioArctic, Biogen, JOMDD/Shimadzu, Julius Clinical, Lilly, MagQu, Novartis, Ono Pharma, Pharmatrophix, Prothena, Roche Diagnostics and Siemens Healthineers, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program. G.T.-B. and H.C.K. are employees of Janssen Research and Development. The remaining authors declare no competing interests.
Figures




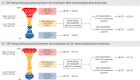



References
-
- McKhann GM, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. J. Alzheimers Assoc. 2011;7:263–269. doi: 10.1016/j.jalz.2011.03.005. - DOI - PMC - PubMed
-
- van Dyck C. H., et al. Lecanemab in early Alzheimer’s disease. N. Engl. J. Med. 388, 9–21 (2023). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

