Acute blood biomarker profiles predict cognitive deficits 6 and 12 months after COVID-19 hospitalization
- PMID: 37653345
- PMCID: PMC10579097
- DOI: 10.1038/s41591-023-02525-y
Acute blood biomarker profiles predict cognitive deficits 6 and 12 months after COVID-19 hospitalization
Abstract
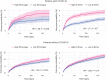
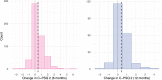
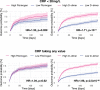
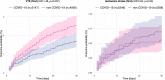
Post-COVID cognitive deficits, including 'brain fog', are clinically complex, with both objective and subjective components. They are common and debilitating, and can affect the ability to work, yet their biological underpinnings remain unknown. In this prospective cohort study of 1,837 adults hospitalized with COVID-19, we identified two distinct biomarker profiles measured during the acute admission, which predict cognitive outcomes 6 and 12 months after COVID-19. A first profile links elevated fibrinogen relative to C-reactive protein with both objective and subjective cognitive deficits. A second profile links elevated D-dimer relative to C-reactive protein with subjective cognitive deficits and occupational impact. This second profile was mediated by fatigue and shortness of breath. Neither profile was significantly mediated by depression or anxiety. Results were robust across secondary analyses. They were replicated, and their specificity to COVID-19 tested, in a large-scale electronic health records dataset. These findings provide insights into the heterogeneous biology of post-COVID cognitive deficits.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures







Comment in
-
Predicting post-COVID-19 cognitive deficits.Nat Rev Neurosci. 2023 Nov;24(11):654. doi: 10.1038/s41583-023-00751-4. Nat Rev Neurosci. 2023. PMID: 37770622 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

