Mitochondrial phospholipid metabolism in health and disease
- PMID: 37655851
- PMCID: PMC10482392
- DOI: 10.1242/jcs.260857
Mitochondrial phospholipid metabolism in health and disease
Abstract
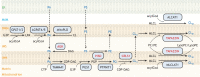
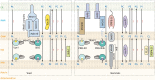
Studies of rare human genetic disorders of mitochondrial phospholipid metabolism have highlighted the crucial role that membrane phospholipids play in mitochondrial bioenergetics and human health. The phospholipid composition of mitochondrial membranes is highly conserved from yeast to humans, with each class of phospholipid performing a specific function in the assembly and activity of various mitochondrial membrane proteins, including the oxidative phosphorylation complexes. Recent studies have uncovered novel roles of cardiolipin and phosphatidylethanolamine, two crucial mitochondrial phospholipids, in organismal physiology. Studies on inter-organellar and intramitochondrial phospholipid transport have significantly advanced our understanding of the mechanisms that maintain mitochondrial phospholipid homeostasis. Here, we discuss these recent advances in the function and transport of mitochondrial phospholipids while describing their biochemical and biophysical properties and biosynthetic pathways. Additionally, we highlight the roles of mitochondrial phospholipids in human health by describing the various genetic diseases caused by disruptions in their biosynthesis and discuss advances in therapeutic strategies for Barth syndrome, the best-studied disorder of mitochondrial phospholipid metabolism.
Keywords: Barth syndrome; Cardiolipin; Membranes; Mitochondria; Phosphatidylethanolamine; Phospholipids.
© 2023. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures

Similar articles
-
Phospholipid ebb and flow makes mitochondria go.J Cell Biol. 2020 Aug 3;219(8):e202003131. doi: 10.1083/jcb.202003131. J Cell Biol. 2020. PMID: 32614384 Free PMC article. Review.
-
Mitochondrial phospholipid transport: Role of contact sites and lipid transport proteins.Prog Lipid Res. 2024 Apr;94:101268. doi: 10.1016/j.plipres.2024.101268. Epub 2024 Jan 7. Prog Lipid Res. 2024. PMID: 38195013 Review.
-
Role for two conserved intermembrane space proteins, Ups1p and Ups2p, [corrected] in intra-mitochondrial phospholipid trafficking.J Biol Chem. 2012 May 4;287(19):15205-18. doi: 10.1074/jbc.M111.338665. Epub 2012 Mar 7. J Biol Chem. 2012. PMID: 22403410 Free PMC article.
-
The role of nonbilayer phospholipids in mitochondrial structure and function.FEBS Lett. 2018 Apr;592(8):1273-1290. doi: 10.1002/1873-3468.12887. Epub 2017 Nov 9. FEBS Lett. 2018. PMID: 29067684 Free PMC article. Review.
-
Lipid metabolism in mitochondrial membranes.J Inherit Metab Dis. 2015 Jan;38(1):137-44. doi: 10.1007/s10545-014-9748-x. Epub 2014 Aug 1. J Inherit Metab Dis. 2015. PMID: 25082432 Review.
Cited by
-
Alterations in Lipid Saturation Trigger Remodeling of the Outer Mitochondrial Membrane.bioRxiv [Preprint]. 2025 Jan 23:2025.01.20.633997. doi: 10.1101/2025.01.20.633997. bioRxiv. 2025. Update in: Mol Biol Cell. 2025 Jul 30:mbcE25010033. doi: 10.1091/mbc.E25-01-0033. PMID: 39896499 Free PMC article. Updated. Preprint.
-
Triacylglycerol mobilization underpins mitochondrial stress recovery.Nat Cell Biol. 2025 Feb;27(2):298-308. doi: 10.1038/s41556-024-01586-6. Epub 2025 Jan 8. Nat Cell Biol. 2025. PMID: 39779944 Free PMC article.
-
Mitochondrial Dysfunction and Atherosclerosis: The Problem and the Search for Its Solution.Biomedicines. 2025 Apr 15;13(4):963. doi: 10.3390/biomedicines13040963. Biomedicines. 2025. PMID: 40299559 Free PMC article. Review.
-
Proteolytic regulation of mitochondrial magnesium channel by m-AAA protease and prohibitin complex.Genetics. 2025 Feb 5;229(2):iyae203. doi: 10.1093/genetics/iyae203. Genetics. 2025. PMID: 39657011
-
Phospholipid and glycerolipid metabolism as potential diagnostic biomarkers for acute pancreatitis.Lipids Health Dis. 2024 Jul 23;23(1):223. doi: 10.1186/s12944-024-02217-7. Lipids Health Dis. 2024. PMID: 39044297 Free PMC article.
References
-
- Achleitner, G., Gaigg, B., Krasser, A., Kainersdorfer, E., Kohlwein, S. D., Perktold, A., Zellnig, G. and Daum, G. (1999). Association between the endoplasmic reticulum and mitochondria of yeast facilitates interorganelle transport of phospholipids through membrane contact. Eur. J. Biochem. 264, 545-553. 10.1046/j.1432-1327.1999.00658.x - DOI - PubMed
-
- Adachi, Y., Itoh, K., Yamada, T., Cerveny, K. L., Suzuki, T. L., Macdonald, P., Frohman, M. A., Ramachandran, R., Iijima, M. and Sesaki, H. (2016). Coincident phosphatidic acid interaction restrains Drp1 in mitochondrial division. Mol. Cell 63, 1034-1043. 10.1016/j.molcel.2016.08.013 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

