Butyrate enhances Clostridioides difficile sporulation in vitro
- PMID: 37655912
- PMCID: PMC10521354
- DOI: 10.1128/jb.00138-23
Butyrate enhances Clostridioides difficile sporulation in vitro
Abstract
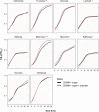
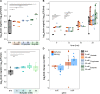
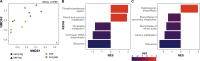
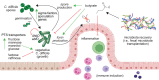
Short-chain fatty acids (SCFAs) are products of bacterial fermentation that help maintain important gut functions such as maintenance of the intestinal barrier, cell signaling, and immune homeostasis. The main SCFAs acetate, propionate, and butyrate have demonstrated beneficial effects for the host, including its importance in alleviating infections caused by pathogens such as Clostridioides difficile. Despite the potential role of SCFAs in mitigating C. difficile infection, their direct effect on C. difficile remains unclear. Through a set of in vitro experiments, we investigated how SCFAs influence C. difficile growth, sporulation, and toxin production. Similar to previous studies, we observed that butyrate decreased growth of C. difficile strain 630 in a dose-dependent manner. The presence of butyrate also increased C. difficile sporulation, with minimal increases in toxin production. RNA-Seq analysis validated our experimental results, demonstrating increased expression of sporulation-related genes in conjunction with changes in metabolic and regulatory genes, such as a putative carbon starvation protein, CstA. Collectively, these data suggest that butyrate may induce alternative C. difficile survival pathways, modifying its growth ability and virulence to persist in the gut environment. IMPORTANCE Several studies suggest that butyrate may modulate gut infections, such as reducing inflammation caused by the healthcare-associated Clostridioides difficile. While studies in both animal models and human studies correlate high levels of butyrate with reduced C. difficile burden, the direct impact of butyrate on C. difficile remains unclear. Our study demonstrates that butyrate directly influences C. difficile by increasing its sporulation and modifying its metabolism, potentially using butyrate as a biomarker to shift survival strategies in a changing gut environment. These data point to additional therapeutic approaches to combat C. difficile in a butyrate-directed manner.
Keywords: Clostridioides difficile; butyrate; growth assay; metabolism; sporulation.
Conflict of interest statement
A.M.S. has consulted with Ferring Pharmaceuticals.
Figures



Update of
-
Butyrate enhances Clostridioides difficile sporulation in vitro.bioRxiv [Preprint]. 2023 Apr 27:2023.04.27.538596. doi: 10.1101/2023.04.27.538596. bioRxiv. 2023. Update in: J Bacteriol. 2023 Sep 26;205(9):e0013823. doi: 10.1128/jb.00138-23. PMID: 37163089 Free PMC article. Updated. Preprint.
References
-
- Guh AY, Mu Y, Winston LG, Johnston H, Olson D, Farley MM, Wilson LE, Holzbauer SM, Phipps EC, Dumyati GK, Beldavs ZG, Kainer MA, Karlsson M, Gerding DN, McDonald LC, Emerging Infections Program Clostridioides difficile Infection Working Group . 2020. Trends in U.S. burden of Clostridioides difficile infection and outcomes. N Engl J Med 382:1320–1330. doi:10.1056/NEJMoa1910215 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

