The Origin of the Intracellular Silver in Bacteria: A Comprehensive Study using Targeting Gold-Silver Alloy Nanoparticles
- PMID: 37661312
- PMCID: PMC11469222
- DOI: 10.1002/adhm.202302084
The Origin of the Intracellular Silver in Bacteria: A Comprehensive Study using Targeting Gold-Silver Alloy Nanoparticles
Abstract
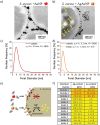
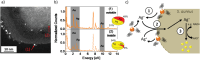
The bactericidal effects of silver nanoparticles (Ag NPs) against infectious strains of multiresistant bacteria is a well-studied phenomenon, highly relevant for many researchers and clinicians battling bacterial infections. However, little is known about the uptake of the Ag NPs into the bacteria, the related uptake mechanisms, and how they are connected to antimicrobial activity. Even less information is available on AgAu alloy NPs uptake. In this work, the interactions between colloidal silver-gold alloy nanoparticles (AgAu NPs) and Staphylococcus aureus (S. aureus) using advanced electron microscopy methods are studied. The localization of the nanoparticles is monitored on the membrane and inside the bacterial cells and the elemental compositions of intra- and extracellular nanoparticle species. The findings reveal the formation of pure silver nanoparticles with diameters smaller than 10 nm inside the bacteria, even though those particles are not present in the original colloid. This finding is explained by a local RElease PEnetration Reduction (REPER) mechanism of silver cations emitted from the AgAu nanoparticles, emphasized by the localization of the AgAu nanoparticles on the bacterial membrane by aptamer targeting ligands. These findings can deepen the understanding of the antimicrobial effect of nanosilver, where the microbes are defusing the attacking silver ions via their reduction, and aid in the development of suitable therapeutic approaches.
Keywords: AgAu; LAL; alloy particles; aptamers; biomedicine; nano-bioconjugates.
© 2023 The Authors. Advanced Healthcare Materials published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- a) Li L.‐L., Yin Q., Cheng J., Lu Y., Adv. Healthcare Mater. 2012, 1, 567; - PubMed
- b) Mazloomi‐Rezvani M., Salami‐Kalajahi M., Roghani‐Mamaqani H., Pirayesh A., Appl. Organomet. Chem. 2018, 32, 8;
- c) Indrasekara A. S. D. S., Paladini B. J., Naczynski D. J., Starovoytov V., Moghe P. V., Fabris L., Adv. Healthcare Mater. 2013, 2, 1370. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

