ErbB2 (HER2)-CAR-NK-92 cells for enhanced immunotherapy of metastatic fusion-driven alveolar rhabdomyosarcoma
- PMID: 37662907
- PMCID: PMC10471977
- DOI: 10.3389/fimmu.2023.1228894
ErbB2 (HER2)-CAR-NK-92 cells for enhanced immunotherapy of metastatic fusion-driven alveolar rhabdomyosarcoma
Abstract
Introduction: Metastatic rhabdomyosarcoma (RMS) is a challenging tumor entity that evades conventional treatments and endogenous antitumor immune responses, highlighting the need for novel therapeutic strategies. Applying chimeric antigen receptor (CAR) technology to natural killer (NK) cells may offer safe, effective, and affordable therapies that enhance cancer immune surveillance.
Methods: Here, we assess the efficacy of clinically usable CAR-engineered NK cell line NK-92/5.28.z against ErbB2-positive RMS in vitro and in a metastatic xenograft mouse model.
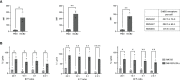
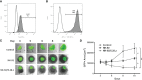
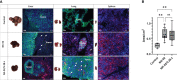
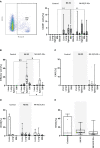
Results: Our results show that NK-92/5.28.z cells effectively kill RMS cells in vitro and significantly prolong survival and inhibit tumor progression in mice. The persistence of NK-92/5.28.z cells at tumor sites demonstrates efficient antitumor response, which could help overcome current obstacles in the treatment of solid tumors.
Discussion: These findings encourage further development of NK-92/5.28.z cells as off-the-shelf immunotherapy for the treatment of metastatic RMS.
Keywords: ERBB2 (HER2/neu); cancer immunotherapy; chimeric antigen receptor; rhabdomyosarcoma; xenograft.
Copyright © 2023 Heim, Moser, Kreyenberg, Bonig, Tonn, Wels, Gradhand, Ullrich, Meister, Koerkamp, Holstege, Drost, Klusmann, Bader, Merker and Rettinger.
Conflict of interest statement
J-HK has advisory roles for Bluebird Bio, Novartis, Roche and Jazz Pharmaceuticals. TT and WW are named as inventors on patents and patent applications related to the study therapeutic owned by their respective academic institutions. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be constructed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

