Stromal cells in the tumor microenvironment: accomplices of tumor progression?
- PMID: 37666813
- PMCID: PMC10477351
- DOI: 10.1038/s41419-023-06110-6
Stromal cells in the tumor microenvironment: accomplices of tumor progression?
Abstract
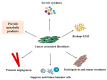
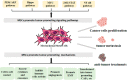
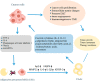
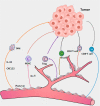
The tumor microenvironment (TME) is made up of cells and extracellular matrix (non-cellular component), and cellular components include cancer cells and non-malignant cells such as immune cells and stromal cells. These three types of cells establish complex signals in the body and further influence tumor genesis, development, metastasis and participate in resistance to anti-tumor therapy. It has attracted scholars to study immune cells in TME due to the significant efficacy of immune checkpoint inhibitors (ICI) and chimeric antigen receptor T (CAR-T) in solid tumors and hematologic tumors. After more than 10 years of efforts, the role of immune cells in TME and the strategy of treating tumors based on immune cells have developed rapidly. Moreover, ICI have been recommended by guidelines as first- or second-line treatment strategies in a variety of tumors. At the same time, stromal cells is another major class of cellular components in TME, which also play a very important role in tumor metabolism, growth, metastasis, immune evasion and treatment resistance. Stromal cells can be recruited from neighboring non-cancerous host stromal cells and can also be formed by transdifferentiation from stromal cells to stromal cells or from tumor cells to stromal cells. Moreover, they participate in tumor genesis, development and drug resistance by secreting various factors and exosomes, participating in tumor angiogenesis and tumor metabolism, regulating the immune response in TME and extracellular matrix. However, with the deepening understanding of stromal cells, people found that stromal cells not only have the effect of promoting tumor but also can inhibit tumor in some cases. In this review, we will introduce the origin of stromal cells in TME as well as the role and specific mechanism of stromal cells in tumorigenesis and tumor development and strategies for treatment of tumors based on stromal cells. We will focus on tumor-associated fibroblasts (CAFs), mesenchymal stem cells (MSCs), tumor-associated adipocytes (CAAs), tumor endothelial cells (TECs) and pericytes (PCs) in stromal cells.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

