Low-glucose culture environment can enhance the wound healing capability of diabetic adipose-derived stem cells
- PMID: 37667384
- PMCID: PMC10478288
- DOI: 10.1186/s13287-023-03478-2
Low-glucose culture environment can enhance the wound healing capability of diabetic adipose-derived stem cells
Abstract
Background: Application of autologous adipose-derived stem cells (ASC) for diabetic chronic wounds has become an emerging treatment option. However, ASCs from diabetic individuals showed impaired cell function and suboptimal wound healing effects. We proposed that adopting a low-glucose level in the culture medium for diabetic ASCs may restore their pro-healing capabilities.
Methods: ASCs from diabetic humans and mice were retrieved and cultured in high-glucose (HG, 4.5 g/L) or low-glucose (LG, 1.0 g/L) conditions. Cell characteristics and functions were investigated in vitro. Moreover, we applied diabetic murine ASCs cultured in HG or LG condition to a wound healing model in diabetic mice to compare their healing capabilities in vivo.
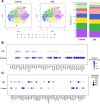
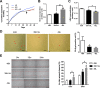
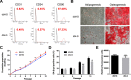
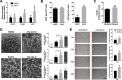
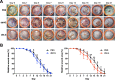
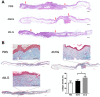
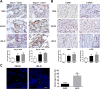
Results: Human ASCs exhibited decreased cell proliferation and migration with enhanced senescence when cultured in HG condition in vitro. Similar findings were noted in ASCs derived from diabetic mice. The inferior cellular functions could be partially recovered when they were cultured in LG condition. In the animal study, wounds healed faster when treated with HG- or LG-cultured diabetic ASCs relative to the control group. Moreover, higher collagen density, more angiogenesis and cellular retention of applied ASCs were found in wound tissues treated with diabetic ASCs cultured in LG condition.
Conclusions: In line with the literature, our study showed that a diabetic milieu exerts an adverse effect on ASCs. Adopting LG culture condition is a simple and effective approach to enhance the wound healing capabilities of diabetic ASCs, which is valuable for the clinical application of autologous ASCs from diabetic patients.
Keywords: Adipose-derived stem cell; Diabetes; Stem cell culture; Stem cell transplantation; Tissue regeneration.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
[Preliminary evaluation and mechanism of adipose-derived stem cell transplantation from allogenic diabetic rats in the treatment of diabetic rat wounds].Zhonghua Shao Shang Za Zhi. 2019 Sep 20;35(9):645-654. doi: 10.3760/cma.j.issn.1009-2587.2019.09.002. Zhonghua Shao Shang Za Zhi. 2019. PMID: 31594182 Chinese.
-
Adipose stem cells isolated from diabetic mice improve cutaneous wound healing in streptozotocin-induced diabetic mice.Stem Cell Res Ther. 2020 Mar 17;11(1):120. doi: 10.1186/s13287-020-01621-x. Stem Cell Res Ther. 2020. PMID: 32183899 Free PMC article.
-
The endothelial cell secretome as a novel treatment to prime adipose-derived stem cells for improved wound healing in diabetes.J Vasc Surg. 2018 Jul;68(1):234-244. doi: 10.1016/j.jvs.2017.05.094. Epub 2017 Jul 29. J Vasc Surg. 2018. PMID: 28760584
-
Adipose-Derived Stem Cells for the Treatment of Diabetic Wound: From Basic Study to Clinical Application.Front Endocrinol (Lausanne). 2022 Jul 11;13:882469. doi: 10.3389/fendo.2022.882469. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35898452 Free PMC article. Review.
-
Adipose-derived Stem Cells for Treatment of Diabetic Foot Ulcers: A Review.Curr Stem Cell Res Ther. 2025;20(5):509-523. doi: 10.2174/011574888X334166240921120502. Curr Stem Cell Res Ther. 2025. PMID: 40525423 Review.
Cited by
-
White adipose tissue in type 2 diabetes and the effect of antidiabetic drugs.Diabetol Metab Syndr. 2025 Apr 4;17(1):116. doi: 10.1186/s13098-025-01678-9. Diabetol Metab Syndr. 2025. PMID: 40186308 Free PMC article. Review.
-
Dual Glyoxalase-1 and β-Klotho Gene-Activated Scaffold Reduces Methylglyoxal and Reprograms Diabetic Adipose-Derived Stem Cells: Prospects in Improved Wound Healing.Pharmaceutics. 2024 Feb 13;16(2):265. doi: 10.3390/pharmaceutics16020265. Pharmaceutics. 2024. PMID: 38399319 Free PMC article.
-
Optimization strategies for mesenchymal stem cell-based analgesia therapy: a promising therapy for pain management.Stem Cell Res Ther. 2024 Jul 18;15(1):211. doi: 10.1186/s13287-024-03828-8. Stem Cell Res Ther. 2024. PMID: 39020426 Free PMC article. Review.
References
-
- Gentile P, Sterodimas A, Pizzicannella J, et al. Systematic review: allogenic use of stromal vascular fraction (SVF) and decellularized extracellular matrices (ECM) as advanced therapy medicinal products (ATMP) in tissue regeneration. Int J Mol Sci. 2020;21(14):4982. doi: 10.3390/ijms21144982. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

