The metabolomic plasma profile of patients with Duchenne muscular dystrophy: providing new evidence for its pathogenesis
- PMID: 37670327
- PMCID: PMC10481483
- DOI: 10.1186/s13023-023-02885-1
The metabolomic plasma profile of patients with Duchenne muscular dystrophy: providing new evidence for its pathogenesis
Abstract
Background: Duchenne muscular dystrophy (DMD) is a fatal genetic muscle-wasting disease that affects 1 in 5000 male births with no current cure. Despite great progress has been made in the research of DMD, its underlying pathological mechanism based on the metabolomics is still worthy of further study. Therefore, it is necessary to gain a deeper understanding of the mechanisms or pathogenesis underlying DMD, which may reveal potential therapeutic targets and/or biomarkers.
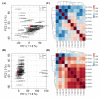
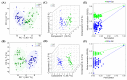
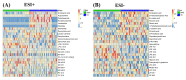
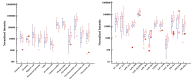
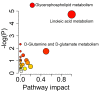
Results: Plasma samples from 42 patients with DMD from a natural history study and 40 age-matched healthy volunteers were subjected to a liquid chromatography-mass spectrometry-based non-targeted metabolomics approach. Acquired metabolic data were evaluated by principal component analysis, partial least squares-discriminant analysis, and metabolic pathway analysis to explore distinctive metabolic patterns in patients with DMD. Differentially expressed metabolites were identified using publicly available and integrated databases. By comparing the DMD and healthy control groups, 25 differential metabolites were detected, including amino acids, unsaturated fatty acids, carnitine, lipids, and metabolites related to the gut microbiota. Correspondingly, linoleic acid metabolism, D-glutamine and D-glutamate metabolism, glycerophospholipid metabolism, and alanine, aspartate, and glutamate metabolism were significantly altered in patients with DMD, compared with those of healthy volunteers.
Conclusions: Our study demonstrated the abnormal metabolism of amino acids, energy, and lipids in patients with DMD, consistent with pathological features, such as recurrent muscle necrosis and regeneration, interstitial fibrosis, and fat replacement. Additionally, we found that metabolites of intestinal flora were disordered in DMD patients, providing support for treatment of intestinal microbia disturbance in DMD diseases. Our study provides a new research strategy for understanding the pathogenesis of DMD.
Keywords: Duchenne muscular dystrophy; Mass spectrometry; Metabonomics; Plasma.
© 2023. Institut National de la Santé et de la Recherche Médicale (INSERM).
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
D- and L-Amino Acid Blood Concentrations Are Affected in Children With Duchenne Muscular Dystrophy.J Cell Mol Med. 2025 May;29(9):e70495. doi: 10.1111/jcmm.70495. J Cell Mol Med. 2025. PMID: 40329488 Free PMC article.
-
Comparative proteomic analyses of Duchenne muscular dystrophy and Becker muscular dystrophy muscles: changes contributing to preserve muscle function in Becker muscular dystrophy patients.J Cachexia Sarcopenia Muscle. 2020 Apr;11(2):547-563. doi: 10.1002/jcsm.12527. Epub 2020 Jan 28. J Cachexia Sarcopenia Muscle. 2020. PMID: 31991054 Free PMC article.
-
Perturbation of muscle metabolism in patients with muscular dystrophy in early or acute phase of disease: In vitro, high resolution NMR spectroscopy based analysis.Clin Chim Acta. 2018 Mar;478:171-181. doi: 10.1016/j.cca.2017.12.036. Epub 2017 Dec 24. Clin Chim Acta. 2018. PMID: 29278724
-
Biomarkers in Duchenne Muscular Dystrophy.Curr Heart Fail Rep. 2022 Apr;19(2):52-62. doi: 10.1007/s11897-022-00541-6. Epub 2022 Apr 7. Curr Heart Fail Rep. 2022. PMID: 35386072 Review.
-
Empowering Muscle Stem Cells for the Treatment of Duchenne Muscular Dystrophy.Cells Tissues Organs. 2022;211(6):641-654. doi: 10.1159/000514305. Epub 2021 Apr 28. Cells Tissues Organs. 2022. PMID: 33910206 Review.
Cited by
-
Characterization of disease-specific alterations in metabolites and effects of mesenchymal stromal cells on dystrophic muscles.Front Cell Dev Biol. 2024 Jun 14;12:1363541. doi: 10.3389/fcell.2024.1363541. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 38946797 Free PMC article.
-
The Role of MicroRNA in the Pathogenesis of Duchenne Muscular Dystrophy.Int J Mol Sci. 2024 Jun 1;25(11):6108. doi: 10.3390/ijms25116108. Int J Mol Sci. 2024. PMID: 38892293 Free PMC article. Review.
-
D- and L-Amino Acid Blood Concentrations Are Affected in Children With Duchenne Muscular Dystrophy.J Cell Mol Med. 2025 May;29(9):e70495. doi: 10.1111/jcmm.70495. J Cell Mol Med. 2025. PMID: 40329488 Free PMC article.
-
How Can Proteomics Help to Elucidate the Pathophysiological Crosstalk in Muscular Dystrophy and Associated Multi-System Dysfunction?Proteomes. 2024 Jan 16;12(1):4. doi: 10.3390/proteomes12010004. Proteomes. 2024. PMID: 38250815 Free PMC article.
-
Beta-hydroxy-beta-methylbutyrate (HMB) improves daily activity and whole-body protein metabolism in Duchenne muscular dystrophy dogs: a pilot study.Sci Rep. 2025 Feb 2;15(1):4026. doi: 10.1038/s41598-025-88651-8. Sci Rep. 2025. PMID: 39894866 Free PMC article.
References
-
- Mercuri E, Bönnemann CG, Muntoni F. Muscular dystrophies. Lancet. 2019;394(10213):2025–38. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

