The proteasome regulates body weight and systemic nutrient metabolism during fasting
- PMID: 37672249
- PMCID: PMC10864006
- DOI: 10.1152/ajpendo.00069.2023
The proteasome regulates body weight and systemic nutrient metabolism during fasting
Abstract
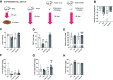
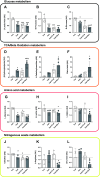
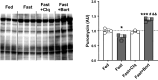
The ubiquitin-proteasome system (UPS) and the autophagy-lysosome pathway are the primary means of degradation in mammalian tissues. We sought to determine the individual contribution of the UPS and autophagy to tissue catabolism during fasting. Mice were overnight fasted for 15 h before regaining food access ("Fed" group, n = 6) or continuing to fast ("Fast" group, n = 7) for 3 h. In addition, to investigate the effects of autophagy on systemic metabolism and tissue degradation, one group of mice was fasted for 18 h and treated with chloroquine ("Fast + CLQ" group, n = 7) and a fourth group of mice was treated with bortezomib ("Fast + Bort" group, n = 7) to assess the contribution of the UPS. Body weight, tissue weight, circulating hormones and metabolites, intracellular signaling pathways, and protein synthesis were investigated. Fasting induced the loss of body weight, liver mass, and white adipose tissue in the Fast and the Fast + CLQ group, whereas the Fast + Bort group maintained tissue and body weight. Fasting reduced glucose and increased β hydroxybutyrate in the circulation of all mice. Both changes were most profound in the Fast + Bort group compared with the other fasting conditions. Molecular signaling indicated a successful inhibition of hepatic UPS with bortezomib and an upregulation of the PI3K/AKT/mTOR pathway. The latter was further supported by an increase in hepatic protein synthesis with bortezomib. Inhibition of the UPS through bortezomib blocks body weight loss and tissue catabolism during an acute overnight fast in mice. The effects were likely mediated through a combined effect of the drug on biomolecule degradation and synthesis.NEW & NOTEWORTHY Bortezomib treatment prevents tissue and body weight loss during fasting. The loss of proteasome activity with bortezomib exacerbates fasting-induced ketogenesis. During fasting, bortezomib increases AMPK and PI3K/AKT signaling in the liver, which promotes protein synthesis.
Keywords: autophagy; fasting; glucose; proteasome; proteolysis.
Conflict of interest statement
M.D.G. reports consulting or advisory roles with Scorpion Therapeutics; stock or other ownership interests in Faeth Therapeutics; honoraria from Novartis, Pfizer, Scorpion Therapeutics; patents, royalties, and other intellectual property with Weill Cornell Medicine. All outside of the scope of this manuscript.
Figures



Similar articles
-
PTEN increases autophagy and inhibits the ubiquitin-proteasome pathway in glioma cells independently of its lipid phosphatase activity.PLoS One. 2013 Dec 13;8(12):e83318. doi: 10.1371/journal.pone.0083318. eCollection 2013. PLoS One. 2013. PMID: 24349488 Free PMC article.
-
mTOR inhibition activates overall protein degradation by the ubiquitin proteasome system as well as by autophagy.Proc Natl Acad Sci U S A. 2015 Dec 29;112(52):15790-7. doi: 10.1073/pnas.1521919112. Epub 2015 Dec 15. Proc Natl Acad Sci U S A. 2015. PMID: 26669439 Free PMC article.
-
Role of the ubiquitin-proteasome system and autophagy in regulation of insulin sensitivity in serum-starved 3T3-L1 adipocytes.Endocr J. 2015;62(8):673-86. doi: 10.1507/endocrj.EJ15-0030. Epub 2015 May 10. Endocr J. 2015. PMID: 25959705
-
Crosstalk between autophagy and proteasome protein degradation systems: possible implications for cancer therapy.Folia Histochem Cytobiol. 2013;51(4):249-64. doi: 10.5603/FHC.2013.0036. Folia Histochem Cytobiol. 2013. PMID: 24497130 Review.
-
The dialogue between the ubiquitin-proteasome system and autophagy: Implications in ageing.Ageing Res Rev. 2020 Dec;64:101203. doi: 10.1016/j.arr.2020.101203. Epub 2020 Oct 29. Ageing Res Rev. 2020. PMID: 33130248 Review.
Cited by
-
AMPK as a mediator of tissue preservation: time for a shift in dogma?Nat Rev Endocrinol. 2024 Sep;20(9):526-540. doi: 10.1038/s41574-024-00992-y. Epub 2024 May 17. Nat Rev Endocrinol. 2024. PMID: 38760482 Review.
-
Primary Roles of Branched Chain Amino Acids (BCAAs) and Their Metabolism in Physiology and Metabolic Disorders.Molecules. 2024 Dec 27;30(1):56. doi: 10.3390/molecules30010056. Molecules. 2024. PMID: 39795113 Free PMC article. Review.
References
-
- Bodine SC, Latres E, Baumhueter S, Lai VK, Nunez L, Clarke BA, Poueymirou WT, Panaro FJ, Na E, Dharmarajan K, Pan ZQ, Valenzuela DM, DeChiara TM, Stitt TN, Yancopoulos GD, Glass DJ. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 294: 1704–1708, 2001. doi:10.1126/science.1065874. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

