Methylene blue accelerates liquid-to-gel transition of tau condensates impacting tau function and pathology
- PMID: 37673952
- PMCID: PMC10482834
- DOI: 10.1038/s41467-023-41241-6
Methylene blue accelerates liquid-to-gel transition of tau condensates impacting tau function and pathology
Abstract
Preventing tau aggregation is a potential therapeutic strategy in Alzheimer's disease and other tauopathies. Recently, liquid-liquid phase separation has been found to facilitate the formation of pathogenic tau conformations and fibrillar aggregates, although many aspects of the conformational transitions of tau during the phase transition process remain unknown. Here, we demonstrate that the tau aggregation inhibitor methylene blue promotes tau liquid-liquid phase separation and accelerates the liquid-to-gel transition of tau droplets independent of the redox activity of methylene blue. We further show that methylene blue inhibits the conversion of tau droplets into fibrils and reduces the cytotoxicity of tau aggregates. Although gelation slows down the mobility of tau and tubulin, it does not impair microtubule assembly within tau droplets. These findings suggest that methylene blue inhibits tau amyloid fibrillization and accelerates tau droplet gelation via distinct mechanisms, thus providing insights into the activity of tau aggregation inhibitors in the context of phase transition.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures

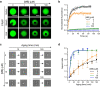
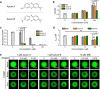

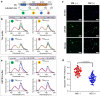

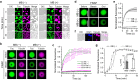
References
-
- Wang Y, Mandelkow E. Tau in physiology and pathology. Nat. Rev. Neurosci. 2016;17:5–21. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

