Iron(III)-based metalloradical catalysis for asymmetric cyclopropanation via a stepwise radical mechanism
- PMID: 37679462
- PMCID: PMC10842623
- DOI: 10.1038/s41557-023-01317-8
Iron(III)-based metalloradical catalysis for asymmetric cyclopropanation via a stepwise radical mechanism
Abstract
Metalloradical catalysis (MRC) exploits the metal-centred radicals present in open-shell metal complexes as one-electron catalysts for the generation of metal-stabilized organic radicals-key intermediates that control subsequent one-electron homolytic reactions. Cobalt(II) complexes of porphyrins, as stable 15e-metalloradicals with a well-defined low-spin d7 configuration, have dominated the ongoing development of MRC. Here, to broaden MRC beyond the use of Co(II)-based metalloradical catalysts, we describe systematic studies that establish the operation of Fe(III)-based MRC and demonstrate an initial application for asymmetric radical transformations. Specifically, we report that five-coordinate iron(III) complexes of porphyrins with an axial ligand, which represent another family of stable 15e-metalloradicals with a d5 configuration, are potent metalloradical catalysts for olefin cyclopropanation with different classes of diazo compounds via a stepwise radical mechanism. This work lays a foundation and mechanistic blueprint for future exploration of Fe(III)-based MRC towards the discovery of diverse stereoselective radical reactions.
© 2023. The Author(s), under exclusive licence to Springer Nature Limited.
Conflict of interest statement
Competing interests
The authors declare no competing interests.
Figures
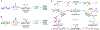




Similar articles
-
Asymmetric C-H Amination via Fe(III)-Metalloradical Catalysis Featuring α-Fe(IV)-Aminyl Radicals as Key Intermediates.J Am Chem Soc. 2025 Jul 9;147(27):24001-24013. doi: 10.1021/jacs.5c07473. Epub 2025 Jun 19. J Am Chem Soc. 2025. PMID: 40534495
-
Metalloradical Catalysis: General Approach for Controlling Reactivity and Selectivity of Homolytic Radical Reactions.Angew Chem Int Ed Engl. 2024 May 13;63(20):e202320243. doi: 10.1002/anie.202320243. Epub 2024 Apr 3. Angew Chem Int Ed Engl. 2024. PMID: 38472114 Free PMC article. Review.
-
Metalloradical Activation of In Situ-Generated α-Alkynyldiazomethanes for Asymmetric Radical Cyclopropanation of Alkenes.J Am Chem Soc. 2022 Feb 9;144(5):2368-2378. doi: 10.1021/jacs.1c13154. Epub 2022 Jan 31. J Am Chem Soc. 2022. PMID: 35099966 Free PMC article.
-
Asymmetric Radical Cyclopropanation of Alkenes with In Situ-Generated Donor-Substituted Diazo Reagents via Co(II)-Based Metalloradical Catalysis.J Am Chem Soc. 2017 Jan 25;139(3):1049-1052. doi: 10.1021/jacs.6b11336. Epub 2017 Jan 12. J Am Chem Soc. 2017. PMID: 28051870 Free PMC article.
-
Iron corrolates: unambiguous chloroiron(III) (corrolate)(2-.) pi-cation radicals.J Inorg Biochem. 2006 Apr;100(4):810-37. doi: 10.1016/j.jinorgbio.2006.01.038. Epub 2006 Mar 7. J Inorg Biochem. 2006. PMID: 16519943 Review.
Cited by
-
Iron Porphyrins and Iron Salens as Highly Enantioselective Catalysts for the Ring-Expansion Reaction of Epoxides to Tetrahydrofurans.Org Lett. 2025 Mar 28;27(12):3083-3088. doi: 10.1021/acs.orglett.5c00767. Epub 2025 Mar 20. Org Lett. 2025. PMID: 40110979 Free PMC article.
-
Chiral iron porphyrin (+)-D4-(por)FeCl catalyzes highly enantioselective cyclopropanation of alkenes using in situ generated diazoacetonitrile with up to 35 000 product turnover.Chem Sci. 2025 Apr 11;16(17):7191-7202. doi: 10.1039/d5sc00461f. eCollection 2025 Apr 30. Chem Sci. 2025. PMID: 40225178 Free PMC article.
-
Catalytic Metalloradical System for Radical 1,6-C(sp3)-H Amination with Concurrent Control of Site-, Chemo-, and Enantio-selectivity.J Am Chem Soc. 2025 May 7;147(18):15755-15766. doi: 10.1021/jacs.5c03259. Epub 2025 Apr 22. J Am Chem Soc. 2025. PMID: 40263670
-
DigiChemTree enables programmable light-induced carbene generation for on demand chemical synthesis.Commun Chem. 2024 Nov 1;7(1):251. doi: 10.1038/s42004-024-01330-z. Commun Chem. 2024. PMID: 39487355 Free PMC article.
-
Organocatalytic enantio- and diastereoselective assembly of cyclopropane-incorporated polycyclic molecules via isobenzopyrylium ions.Chem Sci. 2024 Aug 22;15(37):15274-9. doi: 10.1039/d4sc03746d. Online ahead of print. Chem Sci. 2024. PMID: 39246368 Free PMC article.
References
-
- Zard SZ Radical Reactions in Organic Synthesis (Oxford Chemistry Masters, 2003).
-
- Curran DP, Porter NA & Giese B Stereochemistry of Radical Reactions: Concepts, Guidelines and Synthetic Applications (Wiley, 2008).
-
- Bar G & Parsons AF Stereoselective radical reactions. Chem. Soc. Rev 32, 251–263 (2003). - PubMed
-
- Mondal S. et al. Enantioselective radical reactions using chiral catalysts. Chem. Rev 122, 5842–5976 (2022). - PubMed
-
- Kern N, Plesniak MP, McDouall JJW & Procter DJ Enantioselective cyclizations and cyclization cascades of samarium ketyl radicals. Nat. Chem 9, 1198–1204 (2017). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

