Volatile anesthetics inhibit presynaptic cGMP signaling to depress presynaptic excitability in rat hippocampal neurons
- PMID: 37683886
- PMCID: PMC10772825
- DOI: 10.1016/j.neuropharm.2023.109705
Volatile anesthetics inhibit presynaptic cGMP signaling to depress presynaptic excitability in rat hippocampal neurons
Abstract
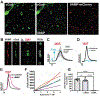
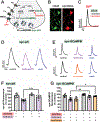
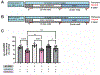
Volatile anesthetics alter presynaptic function through effects on Ca2+ influx and neurotransmitter release. These actions are proposed to play important roles in their pleiotropic neurophysiological effects including immobility, unconsciousness and amnesia. Nitric oxide and cyclic guanosine monophosphate (NO/cGMP) signaling has been implicated in presynaptic mechanisms, and disruption of NO/cGMP signaling has been shown to alter sensitivity to volatile anesthetics in vivo. We investigated volatile anesthetic actions NO/cGMP signaling in relation to presynaptic function in cultured rat hippocampal neurons using pharmacological tools and genetically encoded biosensors and sequestering probes of cGMP levels. Using the fluorescent cGMP biosensor cGull, we found that electrical stimulation-evoked NMDA-type glutamate receptor-independent presynaptic cGMP transients were inhibited 33.2% by isoflurane (0.51 mM) and 26.4% by sevoflurane (0.57 mM) (p < 0.0001) compared to control stimulation without anesthetic. Stimulation-evoked cGMP transients were blocked by the nonselective inhibitor of nitric oxide synthase N-ω-nitro-l-arginine, but not by the selective neuronal nitric oxide synthase inhibitor N5-(1-imino-3-butenyl)-l-ornithine. Isoflurane and sevoflurane inhibition of stimulation-evoked increases in presynaptic Ca2+ concentration, measured with synaptophysin-GCaMP6f, and of synaptic vesicle exocytosis, measured with synaptophysin-pHlourin, was attenuated in neurons expressing the cGMP scavenger protein sponge (inhibition of exocytosis reduced by 54% for isoflurane and by 53% for sevoflurane). The anesthetic-induced reduction in presynaptic excitability was partially occluded by inhibition of HCN channels, a cGMP-modulated excitatory ion channel that can facilitate glutamate release. We propose that volatile anesthetics depress presynaptic cGMP signaling and downstream effectors like HCN channels that are essential to presynaptic function and excitability. These findings identify novel mechanisms by which volatile anesthetics depress synaptic transmission via second messenger signaling involving the NO/cGMP pathway in hippocampal neurons.
Keywords: Cyclic GMP; HCN channel; Isoflurane; Mechanisms of anesthesia; Neurotransmitter release; Nitric oxide; Sevoflurane; Synaptic transmission.
Copyright © 2023 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of competing interest Funding for the project was provided by USNational Institutes of Health Grant R01 GM058055-21 (to HCH) and F31 GM133115 (to VO). IAS, KP, and VO have no competing interests to declare. HCH is editor-in-chief of the British Journal of Anesthesia and has received consulting fees from Elsevier unrelated to this work.
Figures
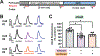
Similar articles
-
α2-Adrenergic Receptor and Isoflurane Modulation of Presynaptic Ca2+ Influx and Exocytosis in Hippocampal Neurons.Anesthesiology. 2016 Sep;125(3):535-46. doi: 10.1097/ALN.0000000000001213. Anesthesiology. 2016. PMID: 27337223 Free PMC article.
-
Inhalational anesthetic effects on rat cerebellar nitric oxide and cyclic guanosine monophosphate production.Anesthesiology. 1997 Mar;86(3):689-98. doi: 10.1097/00000542-199703000-00022. Anesthesiology. 1997. PMID: 9066336
-
Isoflurane Alters Presynaptic Endoplasmic Reticulum Calcium Dynamics in Wild-Type and Malignant Hyperthermia-Susceptible Rodent Hippocampal Neurons.eNeuro. 2023 Aug 29;10(8):ENEURO.0114-23.2023. doi: 10.1523/ENEURO.0114-23.2023. Print 2023 Aug. eNeuro. 2023. PMID: 37591734 Free PMC article.
-
Mechanistic consideration of the effect of perioperative volatile anesthetics on phagocytes.Clin Immunol. 2021 Jan;222:108635. doi: 10.1016/j.clim.2020.108635. Epub 2020 Nov 17. Clin Immunol. 2021. PMID: 33217544 Free PMC article. Review.
-
Mechanism of action of volatile anesthetics: role of protein kinase C.Cell Mol Neurobiol. 2003 Dec;23(6):877-85. doi: 10.1023/b:cemn.0000005316.38838.71. Cell Mol Neurobiol. 2003. PMID: 14964775 Free PMC article. Review.
Cited by
-
The Interplay between cGMP and Calcium Signaling in Alzheimer's Disease.Int J Mol Sci. 2022 Jun 24;23(13):7048. doi: 10.3390/ijms23137048. Int J Mol Sci. 2022. PMID: 35806059 Free PMC article. Review.
-
In vivo imaging reveals a synchronized correlation among neurotransmitter dynamics during propofol and sevoflurane anesthesia.Zool Res. 2024 May 18;45(3):679-690. doi: 10.24272/j.issn.2095-8137.2023.302. Zool Res. 2024. PMID: 38766749 Free PMC article.
References
-
- Hemmings HC Jr., Akabas MH, Goldstein PA, Trudell JR, Orser BA, Harrison NL. Emerging molecular mechanisms of general anesthetic action. Trends Pharmacol Sci 2005; 26: 503–10 - PubMed
-
- Study RE. Isoflurane inhibits multiple voltage-gated calcium currents in hippocampal pyramidal neurons. Anesthesiology 1994; 81: 104–16 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

