Biomarkers of Oxidative Stress in Diabetes Mellitus with Diabetic Nephropathy Complications
- PMID: 37686346
- PMCID: PMC10488183
- DOI: 10.3390/ijms241713541
Biomarkers of Oxidative Stress in Diabetes Mellitus with Diabetic Nephropathy Complications
Abstract
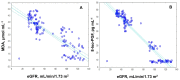
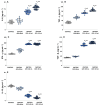
The present study aimed to investigate and compare biomarkers of oxidative stress and the activity of antioxidant enzymes in the plasma of patients with different stages of diabetic nephropathy. For this purpose, we studied (1) the levels of reactive oxygen species and reactive nitrogen species as oxidative stress parameters, (2) lipid and protein oxidation, (3) the activity of antioxidant enzymes, and (4) cytokine production. Patients with type 2 diabetes mellitus were divided into three groups according to the loss of renal function: patients with compensated diabetes mellitus with normal renal function DMT2N0 measured as an estimated glomerular filtration rate (eGFR) ≥ 90 mL/min/1.73 m2, a group with decompensated diabetes mellitus with complication diabetic nephropathy and mild-to-moderate loss of renal function DMT2N1 (eGFR < 60 mL/min/1.73 m2: 59-45 mL/min/1.73 m2), and a decompensated diabetes mellitus with diabetic nephropathy group with moderate-to-severe loss of renal function DMT2N2 (eGFR > 30 mL/min/1.73 m2: 30-44 mL/min/1.73 m2). All results were compared with healthy volunteers. The results showed that patients with diabetic nephropathy had significantly higher levels of ROS, cytokine production, and end products of lipid and protein oxidation compared to healthy volunteers. Furthermore, patients with diabetic nephropathy had depleted levels of nitric oxide (NO), an impaired NO synthase (NOS) system, and reduced antioxidant enzyme activity (p < 0.05). These findings suggest that patients with impaired renal function are unable to compensate for oxidative stress. The decreased levels of NO radicals in patients with advanced renal complications may be attributed to damage NO availability in plasma. The study highlights the compromised oxidative status as a contributing factor to impaired renal function in patients with decompensated type 2 diabetes mellitus. The findings of this study have implications for understanding the pathogenesis of diabetic nephropathy and the role of oxidative stress and chronic inflammation in its development. The assessment of oxidative stress levels and inflammatory biomarkers may aid in the early detection and prediction of diabetic complications.
Keywords: NO radicals; NOS; diabetes mellitus; diabetic nephropathy; oxidative stress.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

