From Monoamine Oxidase Inhibition to Antiproliferative Activity: New Biological Perspectives for Polyamine Analogs
- PMID: 37687158
- PMCID: PMC10490032
- DOI: 10.3390/molecules28176329
From Monoamine Oxidase Inhibition to Antiproliferative Activity: New Biological Perspectives for Polyamine Analogs
Abstract
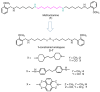
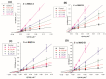
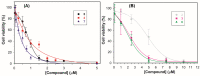
Monoamine oxidases (MAOs) are well-known pharmacological targets in neurological and neurodegenerative diseases. However, recent studies have revealed a new role for MAOs in certain types of cancer such as glioblastoma and prostate cancer, in which they have been found overexpressed. This finding is opening new frontiers for MAO inhibitors as potential antiproliferative agents. In light of our previous studies demonstrating how a polyamine scaffold can act as MAO inhibitor, our aim was to search for novel analogs with greater inhibitory potency for human MAOs and possibly with antiproliferative activity. A small in-house library of polyamine analogs (2-7) was selected to investigate the effect of constrained linkers between the inner amine functions of a polyamine backbone on the inhibitory potency. Compounds 4 and 5, characterized by a dianiline (4) or dianilide (5) moiety, emerged as the most potent, reversible, and mainly competitive MAO inhibitors (Ki < 1 μM). Additionally, they exhibited a high antiproliferative activity in the LN-229 human glioblastoma cell line (GI50 < 1 μM). The scaffold of compound 5 could represent a potential starting point for future development of anticancer agents endowed with MAO inhibitory activity.
Keywords: LN-229 cells; antiproliferative activity; docking studies; inhibitors; monoamine oxidases; polyamine analogs.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Carradori S., Fantacuzzi M., Ammazzalorso A., Angeli A., De Filippis B., Galati S., Petzer A., Petzer J.P., Poli G., Tuccinardi T., et al. Resveratrol Analogues as Dual Inhibitors of Monoamine Oxidase B and Carbonic Anhydrase VII: A New Multi-Target Combination for Neurodegenerative Diseases? Molecules. 2022;27:7816. doi: 10.3390/molecules27227816. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

