Global proteomic identifies multiple cancer-related signaling pathways altered by a gut pathobiont associated with colorectal cancer
- PMID: 37696912
- PMCID: PMC10495336
- DOI: 10.1038/s41598-023-41951-3
Global proteomic identifies multiple cancer-related signaling pathways altered by a gut pathobiont associated with colorectal cancer
Erratum in
-
Author Correction: Global proteomic identifies multiple cancer-related signaling pathways altered by a gut pathobiont associated with colorectal cancer.Sci Rep. 2023 Oct 17;13(1):17676. doi: 10.1038/s41598-023-44834-9. Sci Rep. 2023. PMID: 37848491 Free PMC article. No abstract available.
Abstract
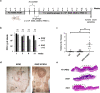
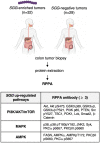
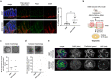
In this work, we investigated the oncogenic role of Streptococcus gallolyticus subsp. gallolyticus (SGG), a gut bacterium associated with colorectal cancer (CRC). We showed that SGG UCN34 accelerates colon tumor development in a chemically induced CRC murine model. Full proteome and phosphoproteome analysis of murine colons chronically colonized by SGG UCN34 revealed that 164 proteins and 725 phosphorylation sites were differentially regulated. Ingenuity Pathway Analysis (IPA) indicates a pro-tumoral shift specifically induced by SGG UCN34, as ~ 90% of proteins and phosphoproteins identified were associated with digestive cancer. Comprehensive analysis of the altered phosphoproteins using ROMA software revealed up-regulation of several cancer hallmark pathways such as MAPK, mTOR and integrin/ILK/actin, affecting epithelial and stromal colonic cells. Importantly, an independent analysis of protein arrays of human colon tumors colonized with SGG showed up-regulation of PI3K/Akt/mTOR and MAPK pathways, providing clinical relevance to our findings. To test SGG's capacity to induce pre-cancerous transformation of the murine colonic epithelium, we grew ex vivo organoids which revealed unusual structures with compact morphology. Taken together, our results demonstrate the oncogenic role of SGG UCN34 in a murine model of CRC associated with activation of multiple cancer-related signaling pathways.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Cancer today. http://gco.iarc.fr/today/home.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

