The CAM lineages of planet Earth
- PMID: 37698538
- PMCID: PMC10799995
- DOI: 10.1093/aob/mcad135
The CAM lineages of planet Earth
Abstract
Background and scope: The growth of experimental studies of crassulacean acid metabolism (CAM) in diverse plant clades, coupled with recent advances in molecular systematics, presents an opportunity to re-assess the phylogenetic distribution and diversity of species capable of CAM. It has been more than two decades since the last comprehensive lists of CAM taxa were published, and an updated survey of the occurrence and distribution of CAM taxa is needed to facilitate and guide future CAM research. We aimed to survey the phylogenetic distribution of these taxa, their diverse morphology, physiology and ecology, and the likely number of evolutionary origins of CAM based on currently known lineages.
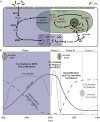
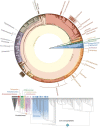
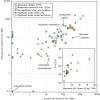
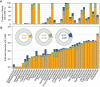
Results and conclusions: We found direct evidence (in the form of experimental or field observations of gas exchange, day-night fluctuations in organic acids, carbon isotope ratios and enzymatic activity) for CAM in 370 genera of vascular plants, representing 38 families. Further assumptions about the frequency of CAM species in CAM clades and the distribution of CAM in the Cactaceae and Crassulaceae bring the currently estimated number of CAM-capable species to nearly 7 % of all vascular plants. The phylogenetic distribution of these taxa suggests a minimum of 66 independent origins of CAM in vascular plants, possibly with dozens more. To achieve further insight into CAM origins, there is a need for more extensive and systematic surveys of previously unstudied lineages, particularly in living material to identify low-level CAM activity, and for denser sampling to increase phylogenetic resolution in CAM-evolving clades. This should allow further progress in understanding the functional significance of this pathway by integration with studies on the evolution and genomics of CAM in its many forms.
Keywords: C3 photosynthesis; C3 + CAM; C4 + CAM; crassulacean acid metabolism; nocturnal acidification; photosynthetic pathway evolution; strong CAM; vascular plants.
© The Author(s) 2023. Published by Oxford University Press on behalf of the Annals of Botany Company. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
Similar articles
-
The diverse diaspora of CAM: a pole-to-pole sketch.Ann Bot. 2023 Nov 25;132(4):597-625. doi: 10.1093/aob/mcad067. Ann Bot. 2023. PMID: 37303205 Free PMC article. Review.
-
CAM photosynthesis: the acid test.New Phytol. 2022 Jan;233(2):599-609. doi: 10.1111/nph.17790. Epub 2021 Nov 5. New Phytol. 2022. PMID: 34637529 Free PMC article.
-
Atmospheric CO2 decline and the timing of CAM plant evolution.Ann Bot. 2023 Nov 25;132(4):753-770. doi: 10.1093/aob/mcad122. Ann Bot. 2023. PMID: 37642245 Free PMC article.
-
Engineering Crassulacean Acid Metabolism in C3 and C4 Plants.Cold Spring Harb Perspect Biol. 2024 Apr 1;16(4):a041674. doi: 10.1101/cshperspect.a041674. Cold Spring Harb Perspect Biol. 2024. PMID: 38052496 Review.
-
Diel rewiring and positive selection of ancient plant proteins enabled evolution of CAM photosynthesis in Agave.BMC Genomics. 2018 Aug 6;19(1):588. doi: 10.1186/s12864-018-4964-7. BMC Genomics. 2018. PMID: 30081833 Free PMC article.
Cited by
-
The diverse diaspora of CAM: a pole-to-pole sketch.Ann Bot. 2023 Nov 25;132(4):597-625. doi: 10.1093/aob/mcad067. Ann Bot. 2023. PMID: 37303205 Free PMC article. Review.
-
Elevated nocturnal respiratory rates in the mitochondria of CAM plants: current knowledge and unanswered questions.Ann Bot. 2023 Nov 25;132(4):855-867. doi: 10.1093/aob/mcad119. Ann Bot. 2023. PMID: 37638861 Free PMC article.
-
Reconciling continuous and discrete models of C4 and CAM evolution.Ann Bot. 2023 Nov 25;132(4):717-725. doi: 10.1093/aob/mcad125. Ann Bot. 2023. PMID: 37675944 Free PMC article.
-
Bringing CAM photosynthesis to the table: Paving the way for resilient and productive agricultural systems in a changing climate.Plant Commun. 2024 Mar 11;5(3):100772. doi: 10.1016/j.xplc.2023.100772. Epub 2023 Nov 21. Plant Commun. 2024. PMID: 37990498 Free PMC article. Review.
-
Low internal air space in plants with crassulacean acid metabolism may be an anatomical spandrel.Ann Bot. 2023 Nov 25;132(4):811-817. doi: 10.1093/aob/mcad109. Ann Bot. 2023. PMID: 37622678 Free PMC article.
References
-
- Akeroyd J, Synge H.. 1992. Higher plant diversity. In: Groombridge B, ed. Global diversity: status of the earth’s living resources. London: Chapman & Hall, 64–87.
-
- Angiosperm Phylogeny Group. 2016. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Botanical Journal of the Linnean Society 181: 1–20.
-
- Arroyo MK, Medina E, Ziegler H.. 1990. Distribution and δ13C values of Portulacaceae species of the High Andes in Northern Chile. Botanica Acta 103: 291–295. doi:10.1111/j.1438-8677.1990.tb00163.x. - DOI
-
- Barbour MM. 2017. Understanding regulation of leaf internal carbon and water transport using online stable isotope techniques. New Phytologist 213: 83–88. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

