INTEGRINS: A BEDSIDE TO BENCH TO BEDSIDE STORY
- PMID: 37701613
- PMCID: PMC10493766
INTEGRINS: A BEDSIDE TO BENCH TO BEDSIDE STORY
Abstract
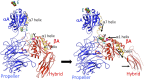
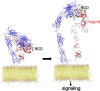
I provide a narrative of the path I took to discover the membrane receptors that mediate leukocyte adhesion, now known as β2 integrins or CD11/CD18. We followed this discovery with the first determination of the 3-D structures of integrins. The latter advance provided the foundation for understanding the unique features of integrins as divalent cation-dependent signaling receptors and as mechanosensitive conduits between the extracellular matrix and the intracellular cytoskeleton. Our structural studies are now opening new paths for taming overactive integrins in disease while minimizing the collateral damage associated with the faulty pharmacodynamics of current integrin inhibitory drugs.
© 2023 The American Clinical and Climatological Association.
Figures
Similar articles
-
The Integrin Receptors: From Discovery to Structure to Medicines.Immunol Rev. 2025 Jan;329(1):e13433. doi: 10.1111/imr.13433. Epub 2024 Dec 26. Immunol Rev. 2025. PMID: 39724488 Review.
-
A role for beta2 integrin (CD11/CD18)-mediated tyrosine signaling in extravasation of human polymorphonuclear neutrophils.Biorheology. 2001;38(2-3):89-100. Biorheology. 2001. PMID: 11381167
-
Human neutrophil adherence to laminin in vitro. Evidence for a distinct neutrophil integrin receptor for laminin.J Exp Med. 1990 Apr 1;171(4):1221-37. doi: 10.1084/jem.171.4.1221. J Exp Med. 1990. PMID: 1969920 Free PMC article.
-
CD11/CD18 leukocyte integrins: new signaling receptors for bacterial endotoxin.J Surg Res. 1997 Nov;73(1):85-9. doi: 10.1006/jsre.1997.5195. J Surg Res. 1997. PMID: 9441798
-
Integrin-mediated calcium signaling and regulation of cell adhesion by intracellular calcium.Bioessays. 1997 Jan;19(1):47-55. doi: 10.1002/bies.950190109. Bioessays. 1997. PMID: 9008416 Review.
Cited by
-
A Novel KGD-Based αIIbβ3 Antagonist Prevents Arterial Thrombosis While Preserving Hemostasis and Avoiding Thrombocytopenia.Int J Mol Sci. 2025 May 9;26(10):4530. doi: 10.3390/ijms26104530. Int J Mol Sci. 2025. PMID: 40429674 Free PMC article.
-
Platelet integrin αIIbβ3 plays a key role in a venous thrombogenesis mouse model.Nat Commun. 2024 Oct 4;15(1):8612. doi: 10.1038/s41467-024-52869-3. Nat Commun. 2024. PMID: 39366965 Free PMC article.
References
-
- Arnaout MA, Pitt J, Cohen HJ, et al. Deficiency of granulocyte-membrane glycoprotein (gp150) in a boy with recurrent bacterial infections. Complement. 1981;306:693. - PubMed
-
- Arnaout MA, Pitt J, Cohen HJ, et al. Deficiency of a granulocyte-membrane glycoprotein (gp150) in a boy with recurrent bacterial infections. N Engl J Med. 1982;306(12):693–9. - PubMed
-
- Crowley CA, Curnutte JT, Rosin RE, et al. An inherited abnormality of neutrophil adhesion. Its genetic transmission and its association with a missing protein. N Engl J Med. 1980;302(21):1163–8. - PubMed
-
- Kohler G, Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975;256:495. - PubMed
-
- Dana N, Pitt J, Todd RF, et al. Deficiency of a monocyte-granulocyte surface glycoprotein Mo1 in man. Clin Res. 1983;31:489.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
