The effect of forskolin and the role of Epac2A during activation, activity, and deactivation of beta cell networks
- PMID: 37701894
- PMCID: PMC10494243
- DOI: 10.3389/fendo.2023.1225486
The effect of forskolin and the role of Epac2A during activation, activity, and deactivation of beta cell networks
Abstract
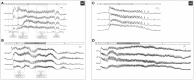
Beta cells couple stimulation by glucose with insulin secretion and impairments in this coupling play a central role in diabetes mellitus. Cyclic adenosine monophosphate (cAMP) amplifies stimulus-secretion coupling via protein kinase A and guanine nucleotide exchange protein 2 (Epac2A). With the present research, we aimed to clarify the influence of cAMP-elevating diterpene forskolin on cytoplasmic calcium dynamics and intercellular network activity, which are two of the crucial elements of normal beta cell stimulus-secretion coupling, and the role of Epac2A under normal and stimulated conditions. To this end, we performed functional multicellular calcium imaging of beta cells in mouse pancreas tissue slices after stimulation with glucose and forskolin in wild-type and Epac2A knock-out mice. Forskolin evoked calcium signals in otherwise substimulatory glucose and beta cells from Epac2A knock-out mice displayed a faster activation. During the plateau phase, beta cells from Epac2A knock-out mice displayed a slightly higher active time in response to glucose compared with wild-type littermates, and stimulation with forskolin increased the active time via an increase in oscillation frequency and a decrease in oscillation duration in both Epac2A knock-out and wild-type mice. Functional network properties during stimulation with glucose did not differ in Epac2A knock-out mice, but the presence of Epac2A was crucial for the protective effect of stimulation with forskolin in preventing a decline in beta cell functional connectivity with time. Finally, stimulation with forskolin prolonged beta cell activity during deactivation, especially in Epac2A knock-out mice.
Keywords: Epac2A KO; amplifying pathway; beta cells; calcium imaging; forskolin; intercellular network; pancreas; tissue slices.
Copyright © 2023 Skelin Klemen, Dolenšek, Križančić Bombek, Pohorec, Gosak, Slak Rupnik and Stožer.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Exendin-4 affects calcium signalling predominantly during activation and activity of beta cell networks in acute mouse pancreas tissue slices.Front Endocrinol (Lausanne). 2024 Jan 16;14:1315520. doi: 10.3389/fendo.2023.1315520. eCollection 2023. Front Endocrinol (Lausanne). 2024. PMID: 38292770 Free PMC article.
-
Pancreatic β-cell response to increased metabolic demand and to pharmacologic secretagogues requires EPAC2A.Diabetes. 2013 Aug;62(8):2796-807. doi: 10.2337/db12-1394. Epub 2013 Apr 11. Diabetes. 2013. PMID: 23578994 Free PMC article.
-
Cooperation between cAMP signalling and sulfonylurea in insulin secretion.Diabetes Obes Metab. 2014 Sep;16 Suppl 1:118-25. doi: 10.1111/dom.12343. Diabetes Obes Metab. 2014. PMID: 25200305 Review.
-
Epac2a-null mice exhibit obesity-prone nature more susceptible to leptin resistance.Int J Obes (Lond). 2017 Feb;41(2):279-288. doi: 10.1038/ijo.2016.208. Epub 2016 Nov 21. Int J Obes (Lond). 2017. PMID: 27867203 Free PMC article.
-
The Role of cAMP in Beta Cell Stimulus-Secretion and Intercellular Coupling.Cells. 2021 Jul 1;10(7):1658. doi: 10.3390/cells10071658. Cells. 2021. PMID: 34359828 Free PMC article. Review.
Cited by
-
Network representation of multicellular activity in pancreatic islets: Technical considerations for functional connectivity analysis.PLoS Comput Biol. 2024 May 13;20(5):e1012130. doi: 10.1371/journal.pcbi.1012130. eCollection 2024 May. PLoS Comput Biol. 2024. PMID: 38739680 Free PMC article.
-
Exendin-4 affects calcium signalling predominantly during activation and activity of beta cell networks in acute mouse pancreas tissue slices.Front Endocrinol (Lausanne). 2024 Jan 16;14:1315520. doi: 10.3389/fendo.2023.1315520. eCollection 2023. Front Endocrinol (Lausanne). 2024. PMID: 38292770 Free PMC article.
References
-
- IDF . Diabetes Atlas. Brussels, Belgium: International Diabetes Federation; (2021).
-
- Ashcroft FM, Ashcroft SJH. Mechanism of insulin secretion. In: Ashcroft FM, Ashcroft SJH, editors. Insulin: molecular biology to pathology. Oxford, UK: IRL Press at Oxford University Press; (1992). p. 97–150.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

