Engineering in vitro models of cystic fibrosis lung disease using neutrophil extracellular trap inspired biomaterials
- PMID: 37701932
- PMCID: PMC10591795
- DOI: 10.1039/d3tb01489d
Engineering in vitro models of cystic fibrosis lung disease using neutrophil extracellular trap inspired biomaterials
Abstract
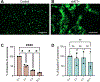
Cystic fibrosis (CF) is a muco-obstructive lung disease where inflammatory responses due to chronic infection result in the accumulation of neutrophil extracellular traps (NETs) in the airways. NETs are web-like complexes comprised mainly of decondensed chromatin that function to capture and kill bacteria. Prior studies have established excess release of NETs in CF airways increases viscoelasticity of mucus secretions and reduces mucociliary clearance. Despite the pivotal role of NETs in CF disease pathogenesis, current in vitro models of this disease do not account for their contribution. Motivated by this, we developed a new approach to study the pathobiological effects of NETs in CF by combining synthetic NET-like biomaterials, composed of DNA and histones, with an in vitro human airway epithelial cell culture model. To determine the impact of synthetic NETs on airway clearance function, we incorporated synthetic NETs into mucin hydrogels and cell culture derived airway mucus to assess their rheological and transport properties. We found that the addition of synthetic NETs significantly increases mucin hydrogel viscoelasticity. As a result, mucociliary transport in vitro was significantly reduced with the addition of mucus containing synthetic NETs. Given the prevalence of bacterial infection in the CF lung, we also evaluated the growth of Pseudomonas aeruginosa in mucus with or without synthetic NETs. We found mucus containing synthetic NETs promoted microcolony growth and prolonged bacterial survival. Together, this work establishes a new biomaterial enabled approach to study innate immunity mediated airway dysfunction in CF.
Conflict of interest statement
Conflicts of interest
The authors have no conflicts to declare.
Figures
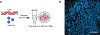

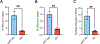

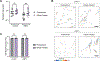
Update of
-
Engineering in vitro models of cystic fibrosis lung disease using neutrophil extracellular trap inspired biomaterials.bioRxiv [Preprint]. 2023 Jun 28:2023.06.26.546583. doi: 10.1101/2023.06.26.546583. bioRxiv. 2023. Update in: J Mater Chem B. 2023 Oct 11;11(39):9419-9430. doi: 10.1039/d3tb01489d. PMID: 37425779 Free PMC article. Updated. Preprint.
Similar articles
-
Engineering in vitro models of cystic fibrosis lung disease using neutrophil extracellular trap inspired biomaterials.bioRxiv [Preprint]. 2023 Jun 28:2023.06.26.546583. doi: 10.1101/2023.06.26.546583. bioRxiv. 2023. Update in: J Mater Chem B. 2023 Oct 11;11(39):9419-9430. doi: 10.1039/d3tb01489d. PMID: 37425779 Free PMC article. Updated. Preprint.
-
Progression of Cystic Fibrosis Lung Disease from Childhood to Adulthood: Neutrophils, Neutrophil Extracellular Trap (NET) Formation, and NET Degradation.Genes (Basel). 2019 Feb 26;10(3):183. doi: 10.3390/genes10030183. Genes (Basel). 2019. PMID: 30813645 Free PMC article. Review.
-
Neutrophil extracellular traps are present in the airways of ENaC-overexpressing mice with cystic fibrosis-like lung disease.BMC Immunol. 2021 Jan 21;22(1):7. doi: 10.1186/s12865-021-00397-w. BMC Immunol. 2021. PMID: 33478382 Free PMC article.
-
Neutrophil extracellular trap (NET)-mediated killing of Pseudomonas aeruginosa: evidence of acquired resistance within the CF airway, independent of CFTR.PLoS One. 2011;6(9):e23637. doi: 10.1371/journal.pone.0023637. Epub 2011 Sep 1. PLoS One. 2011. PMID: 21909403 Free PMC article.
-
Understanding the Entanglement: Neutrophil Extracellular Traps (NETs) in Cystic Fibrosis.Front Cell Infect Microbiol. 2017 Apr 6;7:104. doi: 10.3389/fcimb.2017.00104. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28428948 Free PMC article. Review.
Cited by
-
Optimizing synthetic cystic fibrosis sputum media for growth of non-typeable Haemophilus influenzae.Access Microbiol. 2025 Jun 20;7(6):000979.v3. doi: 10.1099/acmi.0.000979.v3. eCollection 2025. Access Microbiol. 2025. PMID: 40548129 Free PMC article.
-
Synthetic mucus barrier arrays as a nanoparticle formulation screening platform.bioRxiv [Preprint]. 2023 Nov 30:2023.11.29.569212. doi: 10.1101/2023.11.29.569212. bioRxiv. 2023. Update in: RSC Pharm. 2024 Apr 18;1(2):218-226. doi: 10.1039/d3pm00057e. PMID: 38076819 Free PMC article. Updated. Preprint.
-
Synthetic mucus barrier arrays as a nanoparticle formulation screening platform.RSC Pharm. 2024 Apr 18;1(2):218-226. doi: 10.1039/d3pm00057e. eCollection 2024 Jun 18. RSC Pharm. 2024. PMID: 38899149 Free PMC article.
-
Determining the toxicological effects of indoor air pollution on both a healthy and an inflammatory-comprised model of the alveolar epithelial barrier in vitro.Part Fibre Toxicol. 2024 May 17;21(1):25. doi: 10.1186/s12989-024-00584-8. Part Fibre Toxicol. 2024. PMID: 38760786 Free PMC article.
-
Lack of correlation between in vitro and within patient measures of P. aeruginosa biofilms in cystic fibrosis.Heliyon. 2024 Jun 4;10(11):e32424. doi: 10.1016/j.heliyon.2024.e32424. eCollection 2024 Jun 15. Heliyon. 2024. PMID: 38933957 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

