ANO1, CaV1.2, and IP3R form a localized unit of EC-coupling in mouse pulmonary arterial smooth muscle
- PMID: 37702787
- PMCID: PMC10499037
- DOI: 10.1085/jgp.202213217
ANO1, CaV1.2, and IP3R form a localized unit of EC-coupling in mouse pulmonary arterial smooth muscle
Abstract
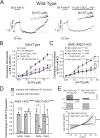
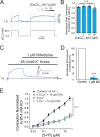
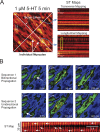
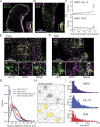
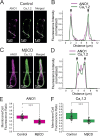
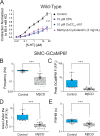
Pulmonary arterial (PA) smooth muscle cells (PASMC) generate vascular tone in response to agonists coupled to Gq-protein receptor signaling. Such agonists stimulate oscillating calcium waves, the frequency of which drives the strength of contraction. These Ca2+ events are modulated by a variety of ion channels including voltage-gated calcium channels (CaV1.2), the Tmem16a or Anoctamin-1 (ANO1)-encoded calcium-activated chloride (CaCC) channel, and Ca2+ release from the sarcoplasmic reticulum through inositol-trisphosphate receptors (IP3R). Although these calcium events have been characterized, it is unclear how these calcium oscillations underly a sustained contraction in these muscle cells. We used smooth muscle-specific ablation of ANO1 and pharmacological tools to establish the role of ANO1, CaV1.2, and IP3R in the contractile and intracellular Ca2+ signaling properties of mouse PA smooth muscle expressing the Ca2+ biosensor GCaMP3 or GCaMP6. Pharmacological block or genetic ablation of ANO1 or inhibition of CaV1.2 or IP3R, or Ca2+ store depletion equally inhibited 5-HT-induced tone and intracellular Ca2+ waves. Coimmunoprecipitation experiments showed that an anti-ANO1 antibody was able to pull down both CaV1.2 and IP3R. Confocal and superresolution nanomicroscopy showed that ANO1 coassembles with both CaV1.2 and IP3R at or near the plasma membrane of PASMC from wild-type mice. We conclude that the stable 5-HT-induced PA contraction results from the integration of stochastic and localized Ca2+ events supported by a microenvironment comprising ANO1, CaV1.2, and IP3R. In this model, ANO1 and CaV1.2 would indirectly support cyclical Ca2+ release events from IP3R and propagation of intracellular Ca2+ waves.
© 2023 Akin et al.
Conflict of interest statement
Disclosures: The authors declare no competing interests exist.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

