Tenofovir diphosphate in dried blood spots and HIV-1 resistance in South Africa
- PMID: 37705102
- PMCID: PMC10500931
- DOI: 10.1186/s12981-023-00552-w
Tenofovir diphosphate in dried blood spots and HIV-1 resistance in South Africa
Abstract
Background: Suboptimal antiretroviral (ART) adherence can lead to virologic failure with consequent HIV-1 resistance. Tenofovir diphosphate (TFV-DP) in dried blood spots (DBS) is a powerful biomarker of cumulative adherence, predictive of future viremia. It has been associated with resistance in Persons With HIV (PWH) in South Africa and the US. We explored the relationship of TFV-DP concentrations with antiretroviral drug resistance at the time of treatment failure in SA.
Methods: Adult PWH from health clinics in Cape Town, South Africa on efavirenz-based first-line ART containing tenofovir disoproxil fumarate (TDF) with an undetectable (< 50 copies/mL) HIV-1 viral load (VL) were prospectively enrolled in an observational cohort for 12 months. Monthly study visits included blood collection for HIV-1 VL and DBS for TFV-DP. The first confirmed viral breakthrough (VB) > 400 copies/mL triggered HIV-1 genotyping at the subsequent visit. An electronic adherence (EA) device monitored ART adherence in real-time, estimated as a percent for the 30-days prior to VB. Wilcoxon rank sum test was used to compare median [IQR] TFV-DP by genotype outcome.
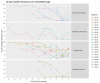
Results: Of 250 individuals, (n = 195, 78% women), 21 experienced VB, with a median of 5 [4;7] months on study, and a median EA of 33.3 [13.3;53.3]%. Demographic characteristics between those with and without VB were similar. Median VL at VB was 4.0 [3.2;4.5] log copies/mL. TFV-DP concentrations trended down towards the VB visit. Median TFV-DP concentrations were significantly higher in those HIV-1 genotype did not amplify due to being virally suppressed at the subsequent visit (n = 10; 380 [227-661] fmol/punch, p = 0.035; EA 45 [24.9; 59.2]%); than in those who were successfully genotyped with evidence of drug resistance (n = 5, 241 [150-247] fmol/punch, EA 20 [6.7;36.7]%) and in individuals who did not have resistance (n = 3, 39.9 [16.6; 93.9] fmol/punch; EA 33.3 [16-38]%). Three genotype collections were not done. Only non-nucleoside reverse transcriptase inhibitor-associated mutations were identified on resistance testing. (K103N, E138K, Y118H).
Conclusion: TFV-DP in DBS showed a step-wise inverse relationship with VB and drug resistance, with evidence of low cumulative ART adherence in PWH who developed antiretroviral resistance. Monitoring TFV-DP concentrations could be a valuable tool for predicting future VB and future resistance.
Keywords: Adherence; Dried blood spots; HIV resistance; South Africa; Tenofovir diphosphate.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
PLA has received consulting fees from Gilead Merck and ViiV and research funding from Gilead paid to his institution.
Figures


References
-
- Castillo-Mancilla JR, Morrow M, Coyle RP, Coleman SS, Gardner EM, Zheng J-H, et al. Tenofovir diphosphate in dried blood spots is strongly associated with viral suppression in individuals with human immunodeficiency virus infections. Clin Infect Dis. 2019;68(8):1335–1342. doi: 10.1093/cid/ciy708. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

