NALIRIFOX versus nab-paclitaxel and gemcitabine in treatment-naive patients with metastatic pancreatic ductal adenocarcinoma (NAPOLI 3): a randomised, open-label, phase 3 trial
- PMID: 37708904
- PMCID: PMC11664154
- DOI: 10.1016/S0140-6736(23)01366-1
NALIRIFOX versus nab-paclitaxel and gemcitabine in treatment-naive patients with metastatic pancreatic ductal adenocarcinoma (NAPOLI 3): a randomised, open-label, phase 3 trial
Abstract
Background: Pancreatic ductal adenocarcinoma remains one of the most lethal malignancies, with few treatment options. NAPOLI 3 aimed to compare the efficacy and safety of NALIRIFOX versus nab-paclitaxel and gemcitabine as first-line therapy for metastatic pancreatic ductal adenocarcinoma (mPDAC).
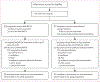
Methods: NAPOLI 3 was a randomised, open-label, phase 3 study conducted at 187 community and academic sites in 18 countries worldwide across Europe, North America, South America, Asia, and Australia. Patients with mPDAC and Eastern Cooperative Oncology Group performance status score 0 or 1 were randomly assigned (1:1) to receive NALIRIFOX (liposomal irinotecan 50 mg/m2, oxaliplatin 60 mg/m2, leucovorin 400 mg/m2, and fluorouracil 2400 mg/m2, administered sequentially as a continuous intravenous infusion over 46 h) on days 1 and 15 of a 28-day cycle or nab-paclitaxel 125 mg/m2 and gemcitabine 1000 mg/m2, administered intravenously, on days 1, 8, and 15 of a 28-day cycle. Balanced block randomisation was stratified by geographical region, performance status, and liver metastases, managed through an interactive web response system. The primary endpoint was overall survival in the intention-to-treat population, evaluated when at least 543 events were observed across the two treatment groups. Safety was evaluated in all patients who received at least one dose of study treatment. This completed trial is registered with ClinicalTrials.gov, NCT04083235.
Findings: Between Feb 19, 2020 and Aug 17, 2021, 770 patients were randomly assigned (NALIRIFOX, 383; nab-paclitaxel-gemcitabine, 387; median follow-up 16·1 months [IQR 13·4-19·1]). Median overall survival was 11·1 months (95% CI 10·0-12·1) with NALIRIFOX versus 9·2 months (8·3-10·6) with nab-paclitaxel-gemcitabine (hazard ratio 0·83; 95% CI 0·70-0·99; p=0·036). Grade 3 or higher treatment-emergent adverse events occurred in 322 (87%) of 370 patients receiving NALIRIFOX and 326 (86%) of 379 patients receiving nab-paclitaxel-gemcitabine; treatment-related deaths occurred in six (2%) patients in the NALIRIFOX group and eight (2%) patients in the nab-paclitaxel-gemcitabine group.
Interpretation: Our findings support use of the NALIRIFOX regimen as a possible reference regimen for first-line treatment of mPDAC.
Funding: Ipsen.
Translation: For the plain language summary see Supplementary Materials section.
Copyright © 2023 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY-NC-ND 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests ZAW has received grants or contracts from Arcus and Bristol Myers Squibb, consulting fees from Amgen, Arcus, Astellas, AstraZeneca, Bayer, Bristol Myers Squibb, Daiichi Sankyo, Gilead, Ipsen, Merck Sharp & Dohme, and Seagen, support for attending meetings or travel from Amgen, Bayer, and Merck Sharp & Dohme, and has participated on a data safety monitoring board or advisory board for Mirati and Pfizer. DM has received grants or contracts from Celgene, Evotec, Incyte, iOnctura, Roche, and Servier, consultancy fees from Incyte, iOnctura, IQVIA, Merck Sharp & Dohme, Servier, and Taiho Pharmaceutical, and has participated on a data safety monitoring board or advisory board for Incyte, Servier, Taiho Pharmaceutical, and Terumo. TM has received grants or contracts (personal) from Merck Sharp & Dohme, Novocure, QED Therapeutics, Roche, Sanofi-Aventis, Servier, and Zymeworks, and received grants or contracts to their institution from AbbVie Farmaceútica, Ability Pharmaceuticals, Agios Pharmaceuticals, Amgen, Aslan Pharmaceuticals, AstraZeneca, Basilea Pharmaceutica International, Bayer, BeiGene, BioKeralty Research Institute, BioLineRx, Blueprint Medicines, Boston Biomedical, Bristol Myers Squibb, Cantargia, Celgene, Eisai, Eli Lilly and Company, Erytech Pharma, FibroGen, Halozyme, Incyte, Ipsen, Loxo Oncology, MedImmune, Merck Sharp & Dohme, Nelum, Novartis, Novocure, OncoMed Pharmaceuticals, QED Therapeutics, Roche, VCN Biosciences, and Zymeworks, payment or honoraria from Janssen and Lilly, support for attending meetings or travel from AstraZeneca, Incyte, Merck Sharp & Dohme, Sanofi, and Servier, and has participated on a data safety monitoring board or advisory board for Ability Pharmaceuticals, AstraZeneca, Basilea Pharma, Baxter, BioLineRX, Celgene, Eisai, Incyte, and Ipsen. RPC has received consulting fees from AstraZeneca, Bayer, Bristol Myers Squibb, Celgene, Eisai, Eli Lilly and Company, Ipsen, Servier, and Roche, honoraria from Astellas, AstraZeneca, Bristol Myers Squibb, Eli Lilly and Company, Roche, and Servier, payment or honoraria from Astellas, AstraZeneca, Bristol Myers Squibb, Eli Lilly and Company, Roche, and Servier, and support for attending meetings or travel from Bristol Myers Squibb, Eli Lilly and Company, Roche, and Servier. SRC has received grants or contracts from AstraZeneca, Exact Sciences, Merck Sharp & Dohme, QED Therapeutics, TerSera Therapeutics, and Zymeworks (personal) and Ipsen (institution), payment or honoraria from Bristol Myers Squibb, Janssen, and Natra (speaker's bureau) and has participated on a data safety monitoring board or advisory board for Daiichi Sankyo. CDLF has received consultancy fees from Bristol Myers Squibb, Daiichi Sankyo, Eisai, Ipsen, Merck Sharp & Dohme, Pierre Fabre Oncologie, and Roche, and support for attending meetings or travel from Merck Sharp & Dohme, Pierre Fabre Oncologie, and Roche. AD has received support for attending meetings or travel from Juniper Biologics. TOG has received grants or contracts from German Research Foundation and Incyte, consulting fees from Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Deciphera, Eli Lilly and Company, Foundation Medicine, MCI, Merck Sharp & Dohme, Novartis, Roche, Sanofi Aventis, and Servier, payment or honoraria from Amgen, Bristol Myers Squibb, Eli Lilly and Company, Merck Sharp & Dohme, Novartis, Roche, Sanofi Aventis, and Servier, has received payment for an expert testimony for Bundesinstitut für Arzneimittel und Medizinprodukte and Daiichi Sankyo, participated on a data safety monitoring board or advisory board for Bayer, Bristol Myers Squibb, Daiichi Sankyo, Eli Lilly and Company, Foundation Medicine, MCI, Merck Sharp & Dohme, Novartis, Roche, Sanofi Aventis, and Servier, and has had a leadership or fiduciary role in board, society, committee, or advocacy group with Arbeitgemeinschaft Internistische Onkologie. EVC has received grants or contracts from Amgen, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly and Company, Ipsen, Merck Sharp & Dohme, Merck KGaA, Novartis, Roche, and Servier, and consulting fees from AbbVie, ALX, Amgen, Array, Astellas, AstraZeneca, Bayer, Beigene, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Eli Lilly and Company, GlaxoSmithKline, Incyte, Ipsen, Merck Sharp & Dohme, Merck KGaA, Mirati, Novartis, Nordic, Pierre Fabre Oncologie, Pfizer, Roche, Seagen, Servier, Takeda, Terumo, Taiho Pharmaceutical, and Zymeworks. ASP has received research funding (personal) from Buzzard Pharmaceuticals, and research funding (institution) from AstraZeneca, Bayer, BioNTech, Bristol Myers Squibb, Camurus, Deciphera, Eli Lilly and Company, Exelixis, G1 Therapeutics, Gilead Sciences, Gritstone Bio, Hutchinson MediPharma, Incyte, Innovative Cellular Therapeutics, Inspirna, Ipsen, ITM Oncologics, Merck Sharp & Dohme, Novartis, NuCana, Relay Therapeutics, Regenxbio, Seagen, SOTIO Biotech, Taiho Pharmaceutical, Tempus, TransThera Biosciences, and Zentalis Pharmaceuticals, had a consulting or advisory role at AADI Bioscience, Advanced Accelerator Applications, Amgen, Astellas, AstraZeneca, Bristol Myers Squibb, Eisai, EMD Serono, Exelixis, Hutchison MediPharma, Incyte, Ipsen, Eli Lilly and Company, Mirati Therapeutics, Novartis, Pfizer, QED Therapeutics, Servier, and Stromatis Pharma, received payment or honoraria from Cardinal Health (honoraria), Ideo Oncology (speaker's bureau), received support for attending meetings or travel from AADI Bioscience, Camurus, Mirati Therapeutics, NuCana, and Pfizer and has stock or stock options in Actinium, Alexion Pharmaceuticals, Aptose Biosciences, and Lynx Health. TB-S has received research funding (institution) from Agios, Arcus, Arys, Atreca, Bayer, Boston Biomedical, Bristol Myers Squibb Eisai, Celgene, Eli Lilly and Company, Ipsen, Clovis, Seagen, Genentech, Novartis, Mirati Therapeutics, Merus, Abgenomics, Incyte, and Pfizer, received royalties from UpToDate, received consultancy fees from Stemline, AbbVie, Aptitude Health, AstraZeneca, Beigene, Blueprint Medicines, Boehringer Ingelheim, Caladrius Biosciences, Celularity, Daiichi Sankyo, Deciphera, Exact Science, Exelixis, Foundation Medicine, GlaxoSmithKline, Illumina, Janssen, Kanaph, MJH Life Sciences, Natera, Sanofi, Sobi, Treos Bio, and Zai Labs (personal), and Arcus, Bayer, Eisai, Genentech, Incyte, Ipsen, Merck Sharp & Dohme, Merck KGA, Merus, Pfizer, Seagen, and Servier (institution), received patents WO/2018/183488 and WO/2019/055687, and has participated on a data safety monitoring board or advisory board for 1Globe, AstraZeneca, Artiva, Eisai, Exelixis, Fibrogen, Immuneering, Imugene, Merck, PanCan, Replimune, Sun Biopharma, Suzhou Kintor, The Valley Hospital, and Xilis. SP has participated on a data safety monitoring board or advisory board for, Askgene Pharma, Boehringer Ingelheim, Ipsen, Janssen, Novartis, and Zymeworks. RAH has received honoraria from Eisai, and participated on a data safety monitoring board or advisory Board at Beigene and Ipsen. ZX, HC, and FB are employed at Ipsen and have stock or stock options in Ipsen. EMO has received grants or contracts from AstraZeneca, Arcus, BioNTech, Elicio, Genentech/Roche, NIH/NCI, Parker Institute, and Pertzye, consulting fees from AstraZeneca, Astellas, Autem, BioNTech, BioSapien, Boehringer Ingelheim, Bristol Myers Squibb, Fibrogen, Ipsen, Merck, Sharp & Dohme, Merus, Neogene, Novartis, Novocure, Tempus, and Thetis (personal), Agios, Eisai, and Genentech/Roche (spouse), has had an uncompensated leadership or fiduciary role in board, society, committee, or advocacy groups with Avner Foundation, National Pancreas Foundation, and Pancreas Cancer Action Network, and has received other financial or non-financial interests from American Association of Cancer Research and American Society of Clinical Oncology. IK and WJL declare no competing interests.
Figures


References
-
- National Institutes of Health Surveillance. National Cancer Institute. Epidemiology and end results program. Cancer stat facts: pancreatic cancer. https://seer.cancer.gov/statfacts/html/pancreas.html (accessed March 1, 2023).
-
- Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011; 364: 1817–25. - PubMed
-
- Goldstein D, El Maraghi RH, Hammel P, et al. Updated survival from a randomized phase III trial (MPACT) of nab-paclitaxel plus gemcitabine versus gemcitabine alone for patients (pts) with metastatic adenocarcinoma of the pancreas. J Clin Oncol 2014; 32 (suppl): 178.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

