Coix seed oil alleviates synovial angiogenesis through suppressing HIF-1α/VEGF-A signaling pathways via SIRT1 in collagen-induced arthritis rats
- PMID: 37715217
- PMCID: PMC10504826
- DOI: 10.1186/s13020-023-00833-6
Coix seed oil alleviates synovial angiogenesis through suppressing HIF-1α/VEGF-A signaling pathways via SIRT1 in collagen-induced arthritis rats
Abstract
Background: Rheumatoid arthritis (RA) is a chronic autoimmune disorder characterized by symmetric arthritis. Coix Seed Oil (CSO) has been shown to reduce inflammation in collagen induced arthritis (CIA) rats. However, the effect of CSO on synovial angiogenesis in RA is unknown. In this study, we aimed to explore whether CSO could inhibit RA synovial angiogenesis and elucidate the underlying mechanisms.
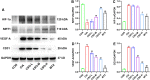
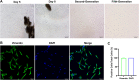
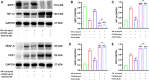
Methods: CIA rat models were established and subjected to different doses of CSO treatments for four weeks in vivo. Arthritis index, paw swelling, and weight were recorded to assess clinical symptoms. Hematoxylin and Eosin staining, Safarnin O fast green staining, Micro-CT, Immunohistochemical, and Immunofluorescence (IF) staining were performed to examined changes in synovial and joint tissues. The serum HIF-1α and VEGF-A levels were evaluated through enzyme-linked immunosorbent assay. Fibroblast-like synoviocytes (FLS) of rats was stimulated with tumor necrosis factor-α (TNF-α) for developing inflammatory model in vitro. Optimal concentrations of CSO and TNF-α for stimulation were measured through Cell Counting Kit-8 test. Wound healing and Transwell migration experiments were employed to determine FLS migratory ability. IF staining was performed to assess HIF-1α nuclear translocation in FLS. Protein levels of SIRT1, HIF-1α, VEGF-A, and CD31 were assessed through Western blot. The isolated aortic rings were induced with recombinant rat VEGF-A 165 (VEGF-A165) to observe the CSO inhibitory impact on angiogenesis ex vivo.
Results: CSO attenuated the progression of arthritis in CIA rats, mitigated histopathological deterioration in synovial and joint tissues, significantly inhibited immature vessels labeled with CD31+/αSMA-, and reduced the micro-vessels in VEGF-A165 induced aortic rings. Moreover, it upregulated SIRT1 protein levels in CIA rats and TNF-α induced FLS, but decreased HIF-1α and VEGF-A protein levels. Furthermore, CSO inhibited the migration ability and HIF-1α nuclear translocation of TNF-α induced FLS. Finally, suppressing SIRT1 levels in TNF-α induced FLS enhanced their migration ability, HIF-1α nuclear translocation, and the protein levels of HIF-1α, VEGF-A, and CD31, whereas the inhibitory effect of CSO on TNF-α induced FLS was severely constrained.
Conclusions: This study indicates that CSO can alleviate synovial angiogenesis through suppressing HIF-1α/VEGF-A signaling pathways via SIRT1 in CIA rats.
Keywords: Angiogenesis; Coix seed oil; HIF-1α; Rheumatoid arthritis; SIRT1.
© 2023. International Society for Chinese Medicine and BioMed Central Ltd.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures









Similar articles
-
Fangji Huangqi Decoction alleviates rheumatoid arthritis through regulating HIF-1α mediated the angiogenesis and the balance between autophagy and apoptosis.J Ethnopharmacol. 2024 Jul 15;329:118061. doi: 10.1016/j.jep.2024.118061. Epub 2024 Apr 16. J Ethnopharmacol. 2024. PMID: 38614265
-
IL-38 restrains inflammatory response of collagen-induced arthritis in rats via SIRT1/HIF-1α signaling pathway.Biosci Rep. 2020 May 29;40(5):BSR20182431. doi: 10.1042/BSR20182431. Biosci Rep. 2020. PMID: 32347300 Free PMC article.
-
Matrine inhibits synovial angiogenesis in collagen-induced arthritis rats by regulating HIF-VEGF-Ang and inhibiting the PI3K/Akt signaling pathway.Mol Immunol. 2022 Jan;141:13-20. doi: 10.1016/j.molimm.2021.11.002. Epub 2021 Nov 12. Mol Immunol. 2022. PMID: 34781187
-
The pathogenesis and regulatory role of HIF-1 in rheumatoid arthritis.Cent Eur J Immunol. 2023;48(4):338-345. doi: 10.5114/ceji.2023.134217. Epub 2024 Jan 8. Cent Eur J Immunol. 2023. PMID: 38558567 Free PMC article. Review.
-
The protective effect of natural medicines in rheumatoid arthritis via inhibit angiogenesis.Front Pharmacol. 2024 May 31;15:1380098. doi: 10.3389/fphar.2024.1380098. eCollection 2024. Front Pharmacol. 2024. PMID: 38881875 Free PMC article. Review.
Cited by
-
Coix Seed Oil Alleviates Hyperuricemia in Mice by Ameliorating Oxidative Stress and Intestinal Microbial Composition.Nutrients. 2025 May 15;17(10):1679. doi: 10.3390/nu17101679. Nutrients. 2025. PMID: 40431419 Free PMC article.
-
Coix Seed Oil Alleviates DSS-Induced Ulcerative Colitis via Intestinal Barrier Repair and Ferroptosis Regulation.J Inflamm Res. 2025 Feb 20;18:2557-2581. doi: 10.2147/JIR.S501745. eCollection 2025. J Inflamm Res. 2025. PMID: 39995826 Free PMC article.
References
-
- Sparks JA, Arthritis R. Rheumatoid arthritis. Ann Intern Med. 2019;170(1):Itc1–Itc16. - PubMed
-
- Park SY, et al. HMGB1 induces angiogenesis in rheumatoid arthritis via HIF-1α activation. Eur J Immunol. 2015;45(4):1216–27. - PubMed
-
- Wang Y, et al. Geniposide downregulates the VEGF/SphK1/S1P pathway and alleviates angiogenesis in rheumatoid arthritis in vivo and in vitro. Phytother Res. 2021;35(8):4347–62. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

