P2X7R influences tau aggregate burden in human tauopathies and shows distinct signalling in microglia and astrocytes
- PMID: 37716378
- PMCID: PMC10896738
- DOI: 10.1016/j.bbi.2023.09.011
P2X7R influences tau aggregate burden in human tauopathies and shows distinct signalling in microglia and astrocytes
Abstract
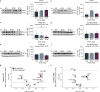
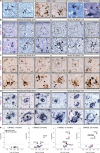
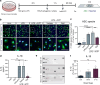
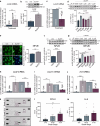
The purinoceptor P2X7R is a promising therapeutic target for tauopathies, including Alzheimer's disease (AD). Pharmacological inhibition or genetic knockdown of P2X7R ameliorates cognitive deficits and reduces pathological tau burden in mice that model aspects of tauopathy, including mice expressing mutant human frontotemporal dementia (FTD)-causing forms of tau. However, disagreements remain over which glial cell types express P2X7R and therefore the mechanism of action is unresolved. Here, we show that P2X7R protein levels increase in human AD post-mortem brain, in agreement with an upregulation of P2RX7 mRNA observed in transcriptome profiles from the AMP-AD consortium. P2X7R protein increases mirror advancing Braak stage and coincide with synapse loss. Using RNAScope we detect P2RX7 mRNA in microglia and astrocytes in human AD brain, including in the vicinity of senile plaques. In cultured microglia, P2X7R activation modulates the NLRP3 inflammasome pathway by promoting the formation of active complexes and release of IL-1β. In astrocytes, P2X7R activates NFκB signalling and increases production of the cytokines CCL2, CXCL1 and IL-6 together with the acute phase protein Lcn2. To further explore the role of P2X7R in a disease-relevant context, we expressed wild-type or FTD-causing mutant forms of tau in mouse organotypic brain slice cultures. Inhibition of P2X7R reduces insoluble tau levels without altering soluble tau phosphorylation or synaptic localisation, suggesting a non-cell autonomous role of glial P2X7R on pathological tau aggregation. These findings support further investigations into the cell-type specific effects of P2X7R-targeting therapies in tauopathies.
Keywords: Alzheimer’s disease; Astrocyte; Human brain; Microglia; P2X(7)R; RNAScope; Tauopathy.
Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
References
-
- Allen M., Carrasquillo M.M., Funk C., Heavner B.D., Zou F., Younkin C.S., Burgess J.D., Chai H.S., Crook J., Eddy J.A., Li H., Logsdon B., Peters M.A., Dang K.K., Wang X., Serie D., Wang C., Nguyen T., Lincoln S., Malphrus K., Bisceglio G., Li M., Golde T.E., Mangravite L.M., Asmann Y., Price N.D., Petersen R.C., Graff-Radford N.R., Dickson D.W., Younkin S.G., Ertekin-Taner N. Human whole genome genotype and transcriptome data for Alzheimer's and other neurodegenerative diseases. Sci. Data. 2016;3 - PMC - PubMed
-
- Anderson C.M., Nedergaard M. Emerging challenges of assigning P2X7 receptor function and immunoreactivity in neurons. Trends Neurosci. 2006;29:257–262. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

