Heatwave resilience of juvenile white sturgeon is associated with epigenetic and transcriptional alterations
- PMID: 37723229
- PMCID: PMC10507091
- DOI: 10.1038/s41598-023-42652-7
Heatwave resilience of juvenile white sturgeon is associated with epigenetic and transcriptional alterations
Abstract
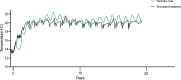
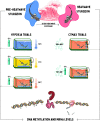
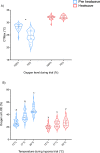
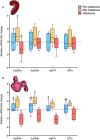
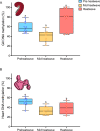
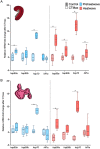
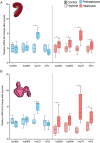
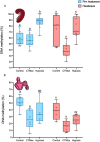
Heatwaves are increasing in frequency and severity, posing a significant threat to organisms globally. In aquatic environments heatwaves are often associated with low environmental oxygen, which is a deadly combination for fish. However, surprisingly little is known about the capacity of fishes to withstand these interacting stressors. This issue is particularly critical for species of extreme conservation concern such as sturgeon. We assessed the tolerance of juvenile white sturgeon from an endangered population to heatwave exposure and investigated how this exposure affects tolerance to additional acute stressors. We measured whole-animal thermal and hypoxic performance and underlying epigenetic and transcriptional mechanisms. Sturgeon exposed to a simulated heatwave had increased thermal tolerance and exhibited complete compensation for the effects of acute hypoxia. These changes were associated with an increase in mRNA levels involved in thermal and hypoxic stress (hsp90a, hsp90b, hsp70 and hif1a) following these stressors. Global DNA methylation was sensitive to heatwave exposure and rapidly responded to acute thermal and hypoxia stress over the course of an hour. These data demonstrate that juvenile white sturgeon exhibit substantial resilience to heatwaves, associated with improved cross-tolerance to additional acute stressors and involving rapid responses in both epigenetic and transcriptional mechanisms.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Stillman JH. Heat waves, the new normal: summertime temperature extremes will impact animals, ecosystems, and human communities. Physiology. 2019;34:86–100. - PubMed
-
- Pörtner Climate change and temperature-dependent biogeography: Oxygen limitation of thermal tolerance in animals. Naturwissenschaften. 2001;88:137–146. - PubMed
-
- Verberk WCEP, Bilton DT, Calosi P, Spicer JI. Oxygen supply in aquatic ectotherms: Partial pressure and solubility together explain biodiversity and size patterns. Ecology. 2011;92:1565–1572. - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources

