Histidine modulates amyloid-like assembly of peptide nanomaterials and confers enzyme-like activity
- PMID: 37726302
- PMCID: PMC10509148
- DOI: 10.1038/s41467-023-41591-1
Histidine modulates amyloid-like assembly of peptide nanomaterials and confers enzyme-like activity
Abstract
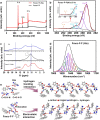
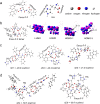
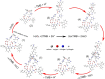
Amyloid-like assembly is not only associated with pathological events, but also leads to the development of novel nanomaterials with unique properties. Herein, using Fmoc diphenylalanine peptide (Fmoc-F-F) as a minimalistic model, we found that histidine can modulate the assembly behavior of Fmoc-F-F and induce enzyme-like catalysis. Specifically, the presence of histidine rearranges the β structure of Fmoc-F-F to assemble nanofilaments, resulting in the formation of active site to mimic peroxidase-like activity that catalyzes ROS generation. A similar catalytic property is also observed in Aβ assembled filaments, which is correlated with the spatial proximity between intermolecular histidine and F-F. Notably, the assembled Aβ filaments are able to induce cellular ROS elevation and damage neuron cells, providing an insight into the pathological relationship between Aβ aggregation and Alzheimer's disease. These findings highlight the potential of histidine as a modulator in amyloid-like assembly of peptide nanomaterials exerting enzyme-like catalysis.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Revisiting the Self-Assembly of Highly Aromatic Phenylalanine Homopeptides.Molecules. 2020 Dec 20;25(24):6037. doi: 10.3390/molecules25246037. Molecules. 2020. PMID: 33419355 Free PMC article. Review.
-
Histidine 14 modulates membrane binding and neurotoxicity of the Alzheimer's disease amyloid-beta peptide.J Alzheimers Dis. 2010;19(4):1387-400. doi: 10.3233/JAD-2010-1334. J Alzheimers Dis. 2010. PMID: 20061603
-
Metal Ions Can Modulate the Self-Assembly and Activity of Catalytic Peptide Amyloids.Langmuir. 2024 Mar 26;40(12):6094-6106. doi: 10.1021/acs.langmuir.3c02983. Epub 2024 Mar 12. Langmuir. 2024. PMID: 38470353
-
Semiconductive and Biocompatible Nanofibrils from the Self-Assembly of Amyloid π-Conjugated Peptides.Biomacromolecules. 2023 Mar 13;24(3):1417-1431. doi: 10.1021/acs.biomac.2c01438. Epub 2023 Feb 27. Biomacromolecules. 2023. PMID: 36847776
-
Elucidating the Structures of Amyloid Oligomers with Macrocyclic β-Hairpin Peptides: Insights into Alzheimer's Disease and Other Amyloid Diseases.Acc Chem Res. 2018 Mar 20;51(3):706-718. doi: 10.1021/acs.accounts.7b00554. Epub 2018 Mar 6. Acc Chem Res. 2018. PMID: 29508987 Free PMC article. Review.
Cited by
-
Peptide nanozymes: An emerging direction for functional enzyme mimics.Bioact Mater. 2024 Sep 4;42:284-298. doi: 10.1016/j.bioactmat.2024.08.033. eCollection 2024 Dec. Bioact Mater. 2024. PMID: 39285914 Free PMC article. Review.
-
Bioinspired ruthenium-manganese-oxygen complex for biocatalytic and radiosensitization therapies to eradicate primary and metastatic tumors.Nat Commun. 2025 Aug 16;16(1):7640. doi: 10.1038/s41467-025-62999-x. Nat Commun. 2025. PMID: 40819134 Free PMC article.
-
CORDAX web server: an online platform for the prediction and 3D visualization of aggregation motifs in protein sequences.Bioinformatics. 2024 May 2;40(5):btae279. doi: 10.1093/bioinformatics/btae279. Bioinformatics. 2024. PMID: 38662570 Free PMC article.
-
Nanotechnology's frontier in combatting infectious and inflammatory diseases: prevention and treatment.Signal Transduct Target Ther. 2024 Feb 21;9(1):34. doi: 10.1038/s41392-024-01745-z. Signal Transduct Target Ther. 2024. PMID: 38378653 Free PMC article. Review.
-
Nanozymes expanding the boundaries of biocatalysis.Nat Commun. 2025 Jul 24;16(1):6817. doi: 10.1038/s41467-025-62063-8. Nat Commun. 2025. PMID: 40707464 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

