Population pharmacokinetics of a combination of miltefosine and paromomycin in Eastern African children and adults with visceral leishmaniasis
- PMID: 37726401
- PMCID: PMC10631828
- DOI: 10.1093/jac/dkad286
Population pharmacokinetics of a combination of miltefosine and paromomycin in Eastern African children and adults with visceral leishmaniasis
Abstract
Objectives: To improve visceral leishmaniasis (VL) treatment in Eastern Africa, 14- and 28-day combination regimens of paromomycin plus allometrically dosed miltefosine were evaluated. As the majority of patients affected by VL are children, adequate paediatric exposure to miltefosine and paromomycin is key to ensuring good treatment response.
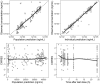
Methods: Pharmacokinetic data were collected in a multicentre randomized controlled trial in VL patients from Kenya, Sudan, Ethiopia and Uganda. Patients received paromomycin (20 mg/kg/day for 14 days) plus miltefosine (allometric dose for 14 or 28 days). Population pharmacokinetic models were developed. Adequacy of exposure and target attainment of paromomycin and miltefosine were evaluated in children and adults.
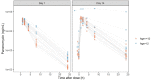
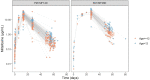
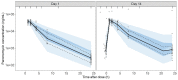
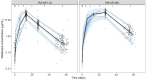
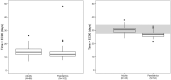
Results: Data from 265 patients (59% ≤12 years) were available for this pharmacokinetic analysis. Paromomycin exposure was lower in paediatric patients compared with adults [median (IQR) end-of-treatment AUC0-24h 187 (162-203) and 242 (217-328) µg·h/mL, respectively], but were both within the IQR of end-of-treatment exposure in Kenyan and Sudanese adult patients from a previous study. Cumulative miltefosine end-of-treatment exposure in paediatric patients and adults [AUCD0-28 517 (464-552) and 524 (456-567) µg·day/mL, respectively] and target attainment [time above the in vitro susceptibility value EC90 27 (25-28) and 30 (28-32) days, respectively] were comparable to previously observed values in adults.
Conclusions: Paromomycin and miltefosine exposure in this new combination regimen corresponded to the desirable levels of exposure, supporting the implementation of the shortened 14 day combination regimen. Moreover, the lack of a clear exposure-response and exposure-toxicity relationship indicated adequate exposure within the therapeutic range in the studied population, including paediatric patients.
© The Author(s) 2023. Published by Oxford University Press on behalf of British Society for Antimicrobial Chemotherapy.
Figures



References
-
- WHO . Global leishmaniasis surveillance: 2019–2020, a baseline for the 2030 roadmap. Weekly epidemiological record. 2021.. https://www.who.int/publications/i/item/who-wer9635-401-419.
-
- Omollo R, Alexander N, Edwards T et al. Safety and efficacy of miltefosine alone and in combination with sodium stibogluconate and liposomal amphotericin B for the treatment of primary visceral leishmaniasis in East Africa: study protocol for a randomized controlled trial. Trials 2011; 12: 166. 10.1186/1745-6215-12-166 - DOI - PMC - PubMed
-
- Wasunna M, Njenga S, Balasegaram M et al. Efficacy and safety of AmBisome in combination with sodium stibogluconate or miltefosine and miltefosine monotherapy for African visceral leishmaniasis: phase II randomized trial. PLoS Negl Trop Dis 2016; 10: e0004880. 10.1371/journal.pntd.0004880 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

