Five-Year Outcome After Continuous Flow LVAD With Full-Magnetic (HeartMate 3) Versus Hybrid Levitation System (HeartWare): A Propensity-Score Matched Study From an All-Comers Multicentre Registry
- PMID: 37727385
- PMCID: PMC10505657
- DOI: 10.3389/ti.2023.11675
Five-Year Outcome After Continuous Flow LVAD With Full-Magnetic (HeartMate 3) Versus Hybrid Levitation System (HeartWare): A Propensity-Score Matched Study From an All-Comers Multicentre Registry
Erratum in
-
Corrigendum: Five-Year Outcome After Continuous Flow LVAD With Full-Magnetic (HeartMate 3) Versus Hybrid Levitation System (HeartWare): A Propensity-Score Matched Study From an All-Comers Multicentre Registry.Transpl Int. 2023 Oct 9;36:12088. doi: 10.3389/ti.2023.12088. eCollection 2023. Transpl Int. 2023. PMID: 37877015 Free PMC article.
Abstract
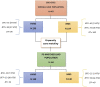
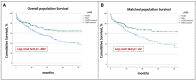
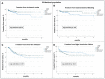
Despite the withdrawal of the HeartWare Ventricular Assist Device (HVAD), hundreds of patients are still supported with this continuous-flow pump, and the long-term management of these patients is still under debate. This study aims to analyse 5 years survival and freedom from major adverse events in patients supported by HVAD and HeartMate3 (HM3). From 2010 to 2022, the MIRAMACS Italian Registry enrolled all-comer patients receiving a LVAD support at seven Cardiac Surgery Centres. Out of 447 LVAD implantation, 214 (47.9%) received HM3 and 233 (52.1%) received HVAD. Cox-regression analysis adjusted for major confounders showed an increased risk for mortality (HR 1.5 [1.2-1.9]; p = 0.031), for both ischemic stroke (HR 2.08 [1.06-4.08]; p = 0.033) and haemorrhagic stroke (HR 2.6 [1.3-4.9]; p = 0.005), and for pump thrombosis (HR 25.7 [3.5-188.9]; p < 0.001) in HVAD patients. The propensity-score matching analysis (130 pairs of HVAD vs. HM3) confirmed a significantly lower 5 years survival (81.25% vs. 64.1%; p 0.02), freedom from haemorrhagic stroke (90.5% vs. 70.1%; p < 0.001) and from pump thrombosis (98.5% vs. 74.7%; p < 0.001) in HVAD cohort. Although similar perioperative outcome, patients implanted with HVAD developed a higher risk for mortality, haemorrhagic stroke and thrombosis during 5 years of follow-up compared to HM3 patients.
Keywords: HeartMate3; HeartWare; continuous-flow LVAD; full-magnetic levitation pump; hybrid levitation system pump.
Copyright © 2023 Francica, Loforte, Attisani, Maiani, Iacovoni, Nisi, Comisso, Terzi, De Bonis, Vendramin, Boffini, Musumeci, Luciani, Rinaldi, Pacini and Onorati.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, et al. ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the European Society of Cardiology (ESC)Developed With the Special Contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J (2016) 37(27):2129–200. 10.1093/eurheartj/ehw128 - DOI - PubMed
-
- FDA Class Irecall for Medtronic HVAD system. Stop New Implants of the Medtronic HVAD System—Letter to Health Care Providers (2021). Available at: https://www.fda.gov/medical-evices/letters-health-care-providers/stop-ne... (Accessed October 16, 2021).
-
- Medtronic. Medtronic Press Release Urgent Medical Device Communication Notification Letter Medtronic HVAD™ System (2021). Available at: https://www.medtronic.com/content/dam/medtronic-com/global/HCP/Documents... (Accessed October 16, 2021).

