Mechanisms of dihydropyridine agonists and antagonists in view of cryo-EM structures of calcium and sodium channels
- PMID: 37728574
- PMCID: PMC10510735
- DOI: 10.1085/jgp.202313418
Mechanisms of dihydropyridine agonists and antagonists in view of cryo-EM structures of calcium and sodium channels
Abstract
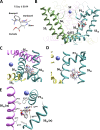
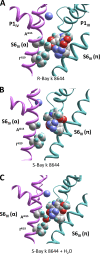
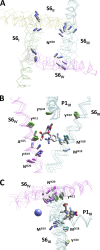
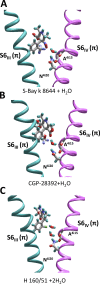
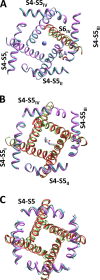
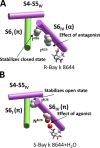
Opposite effects of 1,4-dihydropyridine (DHP) agonists and antagonists on the L-type calcium channels are a challenging problem. Cryo-EM structures visualized DHPs between the pore-lining helices S6III and S6IV in agreement with published mutational data. However, the channel conformations in the presence of DHP agonists and antagonists are virtually the same, and the mechanisms of the ligands' action remain unclear. We docked the DHP agonist S-Bay k 8644 and antagonist R-Bay k 8644 in Cav1.1 channel models with or without π-bulges in helices S6III and S6IV. Cryo-EM structures of the DHP-bound Cav1.1 channel show a π-bulge in helix S6III but not in S6IV. The antagonist's hydrophobic group fits into the hydrophobic pocket formed by residues in S6IV. The agonists' polar NO2 group is too small to fill up the pocket. A water molecule could sterically fit into the void space, but its contacts with isoleucine in helix S6IV (motif INLF) would be unfavorable. In a model with π-bulged S6IV, this isoleucine turns away from the DHP molecule and its position is occupied by the asparagine from the same motif INLF. The asparagine provides favorable contacts for the water molecule at the agonist's NO2 group but unfavorable contacts for the antagonist's methoxy group. In our models, the DHP antagonist stabilizes entirely α-helical S6IV. In contrast, the DHP agonist stabilizes π-bulged helix S6IV whose C-terminal part turned and rearranged the activation-gate region. This would stabilize the open channel. Thus, agonists, but not antagonists, would promote channel opening by stabilizing π-bulged helix S6IV.
© 2023 Tikhonov and Zhorov.
Conflict of interest statement
Disclosures: The authors declare no competing interests exist.
Figures
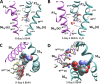
Comment in
-
Elucidating the clandestine behavior of enantiomeric DHPs in calcium channels.J Gen Physiol. 2023 Nov 6;155(11):e202313481. doi: 10.1085/jgp.202313481. Epub 2023 Oct 13. J Gen Physiol. 2023. PMID: 37830978 Free PMC article.
Similar articles
-
Homology model of dihydropyridine receptor: implications for L-type Ca(2+) channel modulation by agonists and antagonists.Arch Biochem Biophys. 2001 Sep 1;393(1):22-41. doi: 10.1006/abbi.2001.2484. Arch Biochem Biophys. 2001. PMID: 11516158
-
Structural Basis of the Modulation of the Voltage-Gated Calcium Ion Channel Cav 1.1 by Dihydropyridine Compounds*.Angew Chem Int Ed Engl. 2021 Feb 8;60(6):3131-3137. doi: 10.1002/anie.202011793. Epub 2020 Dec 10. Angew Chem Int Ed Engl. 2021. PMID: 33125829 Free PMC article.
-
Activation of purified cardiac ryanodine receptors by dihydropyridine agonists.Am J Physiol Heart Circ Physiol. 2001 Mar;280(3):H1201-7. doi: 10.1152/ajpheart.2001.280.3.H1201. Am J Physiol Heart Circ Physiol. 2001. PMID: 11179064
-
Molecular studies on the voltage dependence of dihydropyridine action on L-type Ca2+ channels. Critical involvement of tyrosine residues in motif IIIS6 and IVS6.J Biol Chem. 1997 Oct 3;272(40):24952-60. doi: 10.1074/jbc.272.40.24952. J Biol Chem. 1997. PMID: 9312099
-
Calcium-agonists.J Mol Cell Cardiol. 1987 May;19 Suppl 2:63-75. doi: 10.1016/s0022-2828(87)80005-6. J Mol Cell Cardiol. 1987. PMID: 2442401 Review.
Cited by
-
Elucidating the clandestine behavior of enantiomeric DHPs in calcium channels.J Gen Physiol. 2023 Nov 6;155(11):e202313481. doi: 10.1085/jgp.202313481. Epub 2023 Oct 13. J Gen Physiol. 2023. PMID: 37830978 Free PMC article.
-
Evolution of iGluR ligand specificity, polyamine regulation, and ion selectivity inferred from a placozoan epsilon receptor.Commun Biol. 2025 Jul 3;8(1):994. doi: 10.1038/s42003-025-08402-3. Commun Biol. 2025. PMID: 40610563 Free PMC article.
-
Molecular Mechanisms of L-Type Calcium Channel Dysregulation in Heart Failure.Int J Mol Sci. 2025 Jun 15;26(12):5738. doi: 10.3390/ijms26125738. Int J Mol Sci. 2025. PMID: 40565202 Free PMC article. Review.
References
-
- Brooks, C.L., III, Pettitt M., and Karplus M.. 1985. Structural and energetic effects of truncating long ranged interactions in ionic and polar fluids. J. Chem. Phys. 83:5897–5908. 10.1063/1.449621 - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

