Lipid Nanoparticles Deliver mRNA to the Brain after an Intracerebral Injection
- PMID: 37729550
- PMCID: PMC10760911
- DOI: 10.1021/acs.biochem.3c00371
Lipid Nanoparticles Deliver mRNA to the Brain after an Intracerebral Injection
Abstract
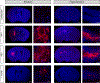
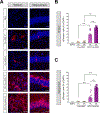
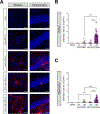
Neurological disorders are often debilitating conditions with no cure. The majority of current therapies are palliative rather than disease-modifying; therefore, new strategies for treating neurological disorders are greatly needed. mRNA-based therapeutics have great potential for treating such neurological disorders; however, challenges with delivery have limited their clinical potential. Lipid nanoparticles (LNPs) are a promising delivery vector for the brain, given their safer toxicity profile and higher efficacy. Despite this, very little is known about LNP-mediated delivery of mRNA into the brain. Here, we employ MC3-based LNPs and successfully deliver Cre mRNA and Cas9 mRNA/Ai9 sgRNA to the adult Ai9 mouse brain; greater than half of the entire striatum and hippocampus was found to be penetrated along the rostro-caudal axis by direct intracerebral injections of MC3 LNP mRNAs. MC3 LNP Cre mRNA successfully transfected cells in the striatum (∼52% efficiency) and hippocampus (∼49% efficiency). In addition, we demonstrate that MC3 LNP Cas9 mRNA/Ai9 sgRNA edited cells in the striatum (∼7% efficiency) and hippocampus (∼3% efficiency). Further analysis demonstrates that MC3 LNPs mediate mRNA delivery to multiple cell types including neurons, astrocytes, and microglia in the brain. Overall, LNP-based mRNA delivery is effective in brain tissue and shows great promise for treating complex neurological disorders.
Conflict of interest statement
Competing interests
The authors declare no competing interests.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

