Spätzle processing enzyme is required to activate dorsal switch protein 1 induced Toll immune signalling pathway in Tenebrio molitor
- PMID: 37733725
- PMCID: PMC10513244
- DOI: 10.1371/journal.pone.0291976
Spätzle processing enzyme is required to activate dorsal switch protein 1 induced Toll immune signalling pathway in Tenebrio molitor
Abstract
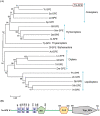
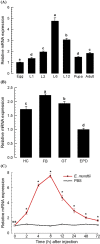
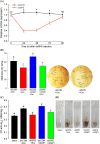
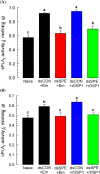
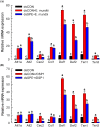
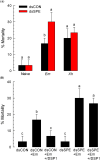
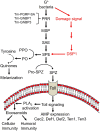
Dorsal switch protein 1 (DSP1) acts as a damage-associated molecular pattern (DAMP) molecule to activate immune responses in Tenebrio molitor. From a previous study in Spodoptera exigua, we found that DSP1 activates Toll immune signalling pathway to induce immune responses by melanisation, PLA2 activity and AMP synthesis. However, the target site of DSP1 in this pathway remains unknown. The objective of this study was to determine the role of spätzle processing enzyme in the DSP1 induced toll immune signalling pathway. To address this, we analyzed spätzle processing enzyme (Tm-SPE) of the three-step serine protease cascade of T. molitor Toll pathway. Tm-SPE expressed in all developmental stages and larval tissues. Upon immune challenge, its expression levels were upregulated but significantly reduced after RNA interference (RNAi). In addition, the induction of immune responses upon immune challenge or recombinant DSP1 injection was significantly increased. Loss of function using RNA interference revealed that the Tm-SPE is involved in connecting DSP1 induced immune responses like hemocyte nodule formation, phenoloxidase (PO) activity, phospholipase A2 (PLA2) activity and antimicrobial peptide (AMP) synthesis. These suggest that Tm-SPE controls the DSP1 induced activation of Toll immune signalling pathway required for both cellular and humoral immune responses. However, to confirm the target molecule of DSP1 in three-step proteolytic cascade, we have to check other upstream serine proteases like Spatzle activating enzyme (SAE) or modular serine protease (MSP).
Copyright: © 2023 Md. Mahi Imam Mollah. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The author have declared that no competing interests exist.
Figures
Similar articles
-
HMGB1-like dorsal switch protein 1 of the mealworm, Tenebrio molitor, acts as a damage-associated molecular pattern.Arch Insect Biochem Physiol. 2021 Jul;107(3):e21795. doi: 10.1002/arch.21795. Epub 2021 May 11. Arch Insect Biochem Physiol. 2021. PMID: 33973266
-
Immune mediation of HMG-like DSP1 via Toll-Spätzle pathway and its specific inhibition by salicylic acid analogs.PLoS Pathog. 2021 Mar 25;17(3):e1009467. doi: 10.1371/journal.ppat.1009467. eCollection 2021 Mar. PLoS Pathog. 2021. PMID: 33765093 Free PMC article.
-
Knockdown of Dorsal switch protein 1 Effect on Growth, Development, and survivability of Tenebrio molitor (Coleoptera: Tenebrionidae).J Invertebr Pathol. 2025 Jul;211:108338. doi: 10.1016/j.jip.2025.108338. Epub 2025 Apr 12. J Invertebr Pathol. 2025. PMID: 40228737
-
Autophagy in Tenebrio molitor Immunity: Conserved Antimicrobial Functions in Insect Defenses.Front Immunol. 2021 May 31;12:667664. doi: 10.3389/fimmu.2021.667664. eCollection 2021. Front Immunol. 2021. PMID: 34135896 Free PMC article. Review.
-
Current Status of Immune Deficiency Pathway in Tenebrio molitor Innate Immunity.Front Immunol. 2022 Jul 4;13:906192. doi: 10.3389/fimmu.2022.906192. eCollection 2022. Front Immunol. 2022. PMID: 35860244 Free PMC article. Review.
Cited by
-
Effect of Emi1 gene silencing on the proliferation and invasion of human breast cancer cells.BMC Mol Cell Biol. 2023 Dec 1;24(1):34. doi: 10.1186/s12860-023-00494-1. BMC Mol Cell Biol. 2023. PMID: 38041032 Free PMC article.
-
Regulation of antimicrobial peptides in Hermetia illucens in response to fungal exposure.Sci Rep. 2024 Nov 28;14(1):29561. doi: 10.1038/s41598-024-80133-7. Sci Rep. 2024. PMID: 39609510 Free PMC article.
-
Developmental transcriptomics reveals stage-specific immune gene expression profiles in Spodoptera frugiperda.Sci Rep. 2025 Jul 11;15(1):25044. doi: 10.1038/s41598-025-06939-1. Sci Rep. 2025. PMID: 40646005 Free PMC article.
-
Ligands of HMG-like dorsal switch protein 1 of Spodoptera exigua leads to mortality in diamondback moth, Plutellaxylostella.Heliyon. 2024 Mar 9;10(6):e27090. doi: 10.1016/j.heliyon.2024.e27090. eCollection 2024 Mar 30. Heliyon. 2024. PMID: 38509914 Free PMC article.
-
Unveiling the Multifaceted Role of HP6: A Critical Regulator of Humoral Immunity in Antheraea pernyi (Lepidoptera: Saturniidae).Int J Mol Sci. 2025 May 9;26(10):4514. doi: 10.3390/ijms26104514. Int J Mol Sci. 2025. PMID: 40429662 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

