Trimodality therapy versus perioperative chemotherapy in the management of locally advanced adenocarcinoma of the oesophagus and oesophagogastric junction (Neo-AEGIS): an open-label, randomised, phase 3 trial
- PMID: 37734399
- PMCID: PMC10567579
- DOI: 10.1016/S2468-1253(23)00243-1
Trimodality therapy versus perioperative chemotherapy in the management of locally advanced adenocarcinoma of the oesophagus and oesophagogastric junction (Neo-AEGIS): an open-label, randomised, phase 3 trial
Abstract
Background: The optimum curative approach to adenocarcinoma of the oesophagus and oesophagogastric junction is unknown. We aimed to compare trimodality therapy (preoperative radiotherapy with carboplatin plus paclitaxel [CROSS regimen]) with optimum contemporaneous perioperative chemotherapy regimens (epirubicin plus cisplatin or oxaliplatin plus fluorouracil or capecitabine [a modified MAGIC regimen] before 2018 and fluorouracil, leucovorin, oxaliplatin, and docetaxel [FLOT] subsequently).
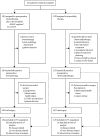
Methods: Neo-AEGIS (CTRIAL-IE 10-14) was an open-label, randomised, phase 3 trial done at 24 centres in Europe. Patients aged 18 years or older with clinical tumour stage T2-3, nodal stage N0-3, and M0 adenocarcinoma of the oesophagus and oesophagogastric junction were randomly assigned to perioperative chemotherapy (three preoperative and three postoperative 3-week cycles of intravenous 50 mg/m2 epirubicin on day 1 plus intravenous 60 mg/m2 cisplatin or intravenous 130 mg/m2 oxaliplatin on day 1 plus continuous infusion of 200 mg/m2 fluorouracil daily or oral 625 mg/m2 capecitabine twice daily up to 2018, with four preoperative and four postoperative 2-week cycles of 2600 mg/m2 fluorouracil, 85 mg/m2 oxaliplatin, 200 mg/m2 leucovorin, and 50 mg/m2 docetaxel intravenously on day 1 as an option from 2018) or trimodality therapy (41·4 Gy in 23 fractions on days 1-5, 8-12, 15-19, 22-26, and 29-31 with intravenous area under the curve 2 mg/mL per min carboplatin plus intravenous 50 mg/m2 paclitaxel on days 1, 8, 15, 22, and 29). The primary endpoint was overall survival, assessed in all randomly assigned patients who received at least one dose of study drug, regardless of which study drug they received, by intention to treat. Secondary endpoints were disease-free survival, site of treatment failure, operative complications, toxicity, pathological response (complete [ypT0N0] and major [tumour regression grade 1 and 2]), margin-free resection (R0), and health-related quality of life. Toxicity and safety data were analysed in the safety population, defined as patients who took at least one dose of study drug, according to treatment actually received. The initial power calculation was based on superiority of trimodality therapy (n=366 patients); it was adjusted after FLOT became an option to a non-inferiority design with a margin of 5% for perioperative chemotherapy (n=540). This study is registered with ClinicalTrials.gov, NCT01726452.
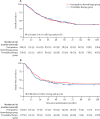
Findings: Between Jan 24, 2013, and Dec 23, 2020, 377 patients were randomly assigned, of whom 362 were included in the intention-to treat population (327 [90%] male and 360 [99%] White): 184 in the perioperative chemotherapy group and 178 in the trimodality therapy group. The trial closed prematurely in December, 2020, after the second interim futility analysis (143 deaths), on the basis of similar survival metrics and the impact of the COVID-19 pandemic. At a median follow-up of 38·8 months (IQR 16·3-55·1), median overall survival was 48·0 months (95% CI 33·6-64·8) in the perioperative chemotherapy group and 49·2 months (34·8-74·4) in the trimodality therapy group (3-year overall survival 55% [95% CI 47-62] vs 57% [49-64]; hazard ratio 1·03 [95% CI 0·77-1·38]; log-rank p=0·82). Median disease-free survival was 32·4 months (95% CI 22·8-64·8) in the perioperative chemotherapy group and 24·0 months (18·0-40·8) in the trimodality therapy group [hazard ratio 0·89 [95% CI 0·68-1·17]; log-rank p=0·41). The pattern of recurrence, locoregional or systemic, was not significantly different (odds ratio 1·35 [95% CI 0·63-2·91], p=0·44). Pathological complete response (odds ratio 0·33 [95% CI 0·14-0·81], p=0·012), major pathological response (0·21 [0·12-0·38], p<0·0001), and R0 rates (0·21 [0·08-0·53], p=0·0003) favoured trimodality therapy. The most common grade 3-4 adverse event was neutropenia (49 [27%] of 183 patients in the perioperative chemotherapy group vs 11 [6%] of 178 patients in the trimodality therapy group), followed by diarrhoea (20 [11%] vs none), and pulmonary embolism (ten [5%] vs nine [5%]). One (1%) patient in the perioperative chemotherapy group and three (2%) patients in the trimodality therapy group died from serious adverse events, two (one in each group) of which were possibly related to treatment. No differences were seen in operative mortality (five [3%] deaths in the perioperative chemotherapy group vs four [2%] in the trimodality therapy group), major morbidity, or in global health status at 1 and 3 years.
Interpretation: Although underpowered and incomplete, Neo-AEGIS provides the largest comprehensive randomised dataset for patients with adenocarcinoma of the oesophagus and oesophagogastric junction treated with perioperative chemotherapy (predominantly the modified MAGIC regimen), and CROSS trimodality therapy, and reports similar 3-year survival and no major differences in operative and health-related quality of life outcomes. We suggest that these data support continued clinical equipoise.
Funding: Health Research Board, Cancer Research UK, Irish Cancer Society, Oesophageal Cancer Fund, and French National Cancer Institute.
Copyright © 2023 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests SRP reports support for the present manuscript from Cancer Research UK; payment or honoraria for lectures, presentations, speaker bureaus, manuscript writing, or educational events from Jabar Al Ahmed Hospital, Kuwait City, Kuwait; support for attending meetings or travel from the Ministry of Health Kuwait. MAL reports payment or honoraria for lectures, presentations, speaker bureaus, manuscript writing or educational events from AstraZeneca; participation on a data safety monitoring board or advisory board for Servier and Agios; other financial or non-financial interests (educational grant to institution from Roche and being principal investigator on clinical trials for MSD, Basilea, Exilexis, Astellas, Daichii Sancho, and Zymeworks). SC reports support for attending meetings or travel from MSD, Pfizer, and Roche. GOG reports payment or honoraria for lectures, presentations, speaker bureaus, manuscript writing, or educational events from AstraZeneca and AbbVie. IP reports support for the present manuscript from Health Research Board, Irish Cancer Society, and Oesophageal Cancer Fund to Cancer Trials Ireland. RR reports consulting fees from Servier; payment or honoraria for educational events from Bristol-Myers Squibb and Servier; support for attending meetings from Servier Laboratories; and advisory board fees from Servier Laboratories. SF reports payment or honoraria for lectures, presentations, speaker bureaus, manuscript writing, or educational events from Servier. GP reports support for the present manuscript from the French National Cancer Institute; grants or contracts from the French National Cancer Institute; consulting fees from Bristol-Myers Squibb, Astellas, and Nestle; payment or honoraria for lectures, presentations, speaker bureaus, manuscript writing, or educational events from the European Society for Medical Oncology; and support for attending meetings or travel from Metronic. DO'T reports honoraria for lectures and speaker bureaus from Ipsen, Novartis, Wyeth Ledrele, and AstraZeneca; support for attending meetings or travel from Ipsen, Novartis, and AstraZeneca; and unpaid leadership roles in other board, society, committee or advocacy groups (European Neuroendocrine Tumour Society Board). RM reports payment or honoraria for lectures, presentations, speaker bureaus, manuscript writing, or educational events from Bayer, Sanofi, Janssen, MSD, Pfizer, Novartis, Clovis, Astellas, Ipsen, and Bristol-Myers Squibb, and support for attending meetings from Pfizer, Janssen, Roche, and Ipsen. All other authors declare no competing interests.
Figures

Comment in
-
Management of locally advanced adenocarcinoma of the oesophagus and oesophagogastric junction: the Neo-AEGIS trial.Lancet Gastroenterol Hepatol. 2024 Feb;9(2):102. doi: 10.1016/S2468-1253(23)00375-8. Lancet Gastroenterol Hepatol. 2024. PMID: 38215771 No abstract available.
-
Management of locally advanced adenocarcinoma of the oesophagus and oesophagogastric junction: the Neo-AEGIS trial.Lancet Gastroenterol Hepatol. 2024 Feb;9(2):102-103. doi: 10.1016/S2468-1253(23)00376-X. Lancet Gastroenterol Hepatol. 2024. PMID: 38215772 No abstract available.
-
Management of locally advanced adenocarcinoma of the oesophagus and oesophagogastric junction: the Neo-AEGIS trial.Lancet Gastroenterol Hepatol. 2024 Feb;9(2):103-104. doi: 10.1016/S2468-1253(23)00407-7. Lancet Gastroenterol Hepatol. 2024. PMID: 38215773 No abstract available.
References
-
- Lagergren J, Smyth E, Cunningham D, Lagergren P. Oesophageal cancer. Lancet. 2017;390:2383–2396. - PubMed
-
- Morgan E, Soerjomataram I, Gavin AT, et al. International trends in oesophageal cancer survival by histological subtype between 1995 and 2014. Gut. 2021;70:234–242. - PubMed
-
- van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–2084. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

