Transition from Dion-Jacobson hybrid layered double perovskites to 1D perovskites for ultraviolet to visible photodetection
- PMID: 37736622
- PMCID: PMC10510777
- DOI: 10.1039/d3sc01919e
Transition from Dion-Jacobson hybrid layered double perovskites to 1D perovskites for ultraviolet to visible photodetection
Abstract
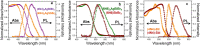
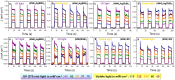
New perovskite phases having diverse optoelectronic properties are the need of the hour. We present five variations of R2AgM(iii)X8, where R = NH3C4H8NH3 (4N4) or NH3C6H12NH3 (6N6); M(iii) = Bi3+ or Sb3+; and X = Br- or I-, by tuning the composition of (4N4)2AgBiBr8, a structurally rich hybrid layered double perovskite (HLDP). (4N4)2AgBiBr8, (4N4)2AgSbBr8, and (6N6)2AgBiBr8 crystallize as Dion-Jacobson (DJ) HLDPs, whereas 1D (6N6)SbBr5, (4N4)-BiI and (4N4)-SbI have trans-connected chains by corner-shared octahedra. Ag+ stays out of the 1D lattice either when SbBr63- distortion is high or if Ag+ needs to octahedrally coordinate with I-. Band structure calculations show a direct bandgap for all the bromide phases except (6N6)2AgBiBr8. (4N4)2AgBiBr8 with lower octahedral tilt shows a maximum UV responsivity of 18.8 ± 0.2 A W-1 and external quantum efficiency (EQE) of 6360 ± 58%, at 2.5 V. When self-powered (0 V), (4N4)-SbI has the best responsivity of 11.7 ± 0.2 mA W-1 under 485 nm visible light, with fast photoresponse ≤100 ms.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



Similar articles
-
New Lead-free Hybrid Layered Double Perovskite Halides: Synthesis, Structural Transition and Ultralow Thermal Conductivity.Angew Chem Int Ed Engl. 2024 Aug 19;63(34):e202406616. doi: 10.1002/anie.202406616. Epub 2024 Jul 4. Angew Chem Int Ed Engl. 2024. PMID: 38771295
-
Structure and Optical Properties of Hybrid-Layered-Double Perovskites (C8H20N2)2AgMBr8 (M = In, Sb, and Bi).Inorg Chem. 2021 Oct 4;60(19):14629-14635. doi: 10.1021/acs.inorgchem.1c01669. Epub 2021 Sep 15. Inorg Chem. 2021. PMID: 34523334
-
Spin Texture Sensitive Photodetection by Dion-Jacobson Tin Halide Perovskites.J Am Chem Soc. 2023 Oct 31. doi: 10.1021/jacs.3c10195. Online ahead of print. J Am Chem Soc. 2023. PMID: 37906676
-
Layered Low-Dimensional Ruddlesden-Popper and Dion-Jacobson Perovskites: From Material Properties to Photovoltaic Device Performance.ChemSusChem. 2023 Oct 6;16(19):e202300736. doi: 10.1002/cssc.202300736. Epub 2023 Aug 1. ChemSusChem. 2023. PMID: 37321966 Review.
-
Additive Engineering for Stable and Efficient Dion-Jacobson Phase Perovskite Solar Cells.Nanomicro Lett. 2023 May 24;15(1):134. doi: 10.1007/s40820-023-01110-9. Nanomicro Lett. 2023. PMID: 37221320 Free PMC article. Review.
Cited by
-
Coordination Polymer Based on a Triangular Carboxylate Core {Fe(μ3-O)(μ-O2CR)6} and an Aliphatic Diamine.Molecules. 2024 May 3;29(9):2125. doi: 10.3390/molecules29092125. Molecules. 2024. PMID: 38731615 Free PMC article.
-
Optoelectronic insights of lead-free layered halide perovskites.Chem Sci. 2024 Apr 23;15(20):7374-7393. doi: 10.1039/d4sc01429d. eCollection 2024 May 22. Chem Sci. 2024. PMID: 38784758 Free PMC article. Review.
References
-
- Tennyson E. M. Doherty T. A. Stranks S. D. Nat. Rev. Mater. 2019;4:573–587. doi: 10.1038/s41578-019-0125-0. - DOI
-
- Guo Z. Jena A. K. Kim G. M. Miyasaka T. Energy Environ. Sci. 2022;15:3171–3222. doi: 10.1039/D2EE00663D. - DOI
-
- Lei H. Hardy D. Gao F. Adv. Funct. Mater. 2021;31:2105898. doi: 10.1002/adfm.202105898. - DOI
-
- Igbari F. Wang Z. K. Liao L. S. Adv. Energy Mater. 2019;9:1803150. doi: 10.1002/aenm.201803150. - DOI
LinkOut - more resources
Full Text Sources

