Increasing Energetic Demands on Photoreceptors in Diabetes Corrects Retinal Lipid Dysmetabolism and Reduces Subsequent Microvascular Damage
- PMID: 37741454
- PMCID: PMC10777362
- DOI: 10.1016/j.ajpath.2023.09.004
Increasing Energetic Demands on Photoreceptors in Diabetes Corrects Retinal Lipid Dysmetabolism and Reduces Subsequent Microvascular Damage
Abstract
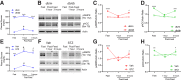
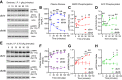
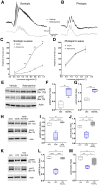
Mechanisms responsible for the pathogenesis of diabetic retinal disease remain incompletely understood, but they likely involve multiple cellular targets, including photoreceptors. Evidence suggests that dysregulated de novo lipogenesis in photoreceptors is a critical early target of diabetes. Following on this observation, the present study aimed to determine whether two interventions shown to improve diabetic retinopathy in mice-pharmacologic visual cycle inhibition and prolonged dark adaptation-reduce photoreceptor anabolic lipid metabolism. Elevated retinal lipid biosynthetic signaling was observed in two mouse models of diabetes, with both models showing reduced retinal AMP-activated kinase (AMPK) signaling, elevated acetyl CoA carboxylase (ACC) signaling, and increased activity of fatty acid synthase, which promotes lipotoxicity in photoreceptors. Although retinal AMPK-ACC axis signaling was dependent on systemic glucose fluctuations in healthy animals, mice with diabetes lacked such regulation. Visual cycle inhibition and prolonged dark adaptation reversed abnormal retinal AMPK-ACC signaling in mice with diabetes. Although visual cycle inhibition reduced the severity of diabetic retinopathy in control mice, as assessed by retinal capillary atrophy, this intervention was ineffective in fatty acid synthase gain-of-function mice. These results suggest that early diabetic retinopathy is characterized by glucose-driven elevations in retinal lipid biosynthetic activity, and that two interventions known to increase photoreceptor glucose demands alleviate disease by reversing these signals.
Copyright © 2023 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Glucose-mediated de novo lipogenesis in photoreceptors drives early diabetic retinopathy.J Biol Chem. 2021 Sep;297(3):101104. doi: 10.1016/j.jbc.2021.101104. Epub 2021 Aug 20. J Biol Chem. 2021. PMID: 34425110 Free PMC article.
-
FGF21 does not require adipocyte AMP-activated protein kinase (AMPK) or the phosphorylation of acetyl-CoA carboxylase (ACC) to mediate improvements in whole-body glucose homeostasis.Mol Metab. 2017 Apr 5;6(6):471-481. doi: 10.1016/j.molmet.2017.04.001. eCollection 2017 Jun. Mol Metab. 2017. PMID: 28580278 Free PMC article.
-
Photoreceptor responses to light in the pathogenesis of diabetic retinopathy.Vis Neurosci. 2020 Sep 14;37:E007. doi: 10.1017/S0952523820000061. Vis Neurosci. 2020. PMID: 32921331 Free PMC article. Review.
-
Photoreceptor Cells Influence Retinal Vascular Degeneration in Mouse Models of Retinal Degeneration and Diabetes.Invest Ophthalmol Vis Sci. 2016 Aug 1;57(10):4272-81. doi: 10.1167/iovs.16-19415. Invest Ophthalmol Vis Sci. 2016. PMID: 27548901 Free PMC article.
-
Modulation of fatty acid metabolism as a potential approach to the treatment of obesity and the metabolic syndrome.Endocrine. 2006 Feb;29(1):91-100. doi: 10.1385/ENDO:29:1:91. Endocrine. 2006. PMID: 16622296 Review.
Cited by
-
Predictive value of triglyceride glucose index and systemic inflammation index for diabetic retinopathy in Type-2 diabetes.Pak J Med Sci. 2025 Apr;41(4):1072-1077. doi: 10.12669/pjms.41.4.11374. Pak J Med Sci. 2025. PMID: 40290244 Free PMC article.
-
Clinical Evidence of a Photoreceptor Origin in Diabetic Retinal Disease.Ophthalmol Sci. 2024 Aug 2;5(1):100591. doi: 10.1016/j.xops.2024.100591. eCollection 2025 Jan-Feb. Ophthalmol Sci. 2024. PMID: 39328824 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

