Improved production of recombinant β-mannanase (TaMan5) in Pichia pastoris and its synergistic degradation of lignocellulosic biomass
- PMID: 37744260
- PMCID: PMC10513448
- DOI: 10.3389/fbioe.2023.1244772
Improved production of recombinant β-mannanase (TaMan5) in Pichia pastoris and its synergistic degradation of lignocellulosic biomass
Abstract
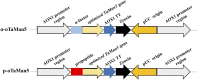
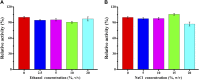
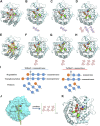
Mannan, a highly abundant and cost-effective natural resource, holds great potential for the generation of high-value compounds such as bioactive polysaccharides and biofuels. In this study, we successfully enhanced the expression of constructed GH5 β-mannanase (TaMan5) from Trichoderma asperellum ND-1 by employing propeptide in Pichia pastoris. By replacing the α-factor with propeptide (MGNRALNSMKFFKSQALALLAATSAVA), TaMan5 activity was significantly increased from 67.5 to 91.7 U/mL. It retained higher activity in the presence of 20% ethanol and 15% NaCl. When incubated with a high concentration of mannotriose or mannotetraose, the transglycosylation action of TaMan5 can be detected, yielding the corresponding production of mannotetraose or mannooligosaccharides. Moreover, the unique mechanism whereby TaMan5 catalyzes the degradation of mannan into mannobiose involves the transglycosylation of mannose to mannotriose or mannotetraose as a substrate to produce a mannotetraose or mannopentose intermediate, respectively. Additionally, the production of soluble sugars from lignocellulose is a crucial step in bioethanol development, and it is noteworthy that TaMan5 could synergistically yield fermentable sugars from corn stover and bagasse. These findings offered valuable insights and strategies for enhancing β-mannanase expression and efficient conversion of lignocellulosic biomass, providing cost-effective and sustainable approaches for high-value biomolecule and biofuel production.
Keywords: Trichoderma asperellum; catalysis mechanism; improved production; synergism; transglycosylation; β-mannanase.
Copyright © 2023 Zheng, Basit, Zhang, Zhuang, Chen and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Biochemical analyses of a novel acidophilic GH5 β-mannanase from Trichoderma asperellum ND-1 and its application in mannooligosaccharides production from galactomannans.Front Microbiol. 2023 Jun 9;14:1191553. doi: 10.3389/fmicb.2023.1191553. eCollection 2023. Front Microbiol. 2023. PMID: 37362936 Free PMC article.
-
Characterization of a novel acidophilic, ethanol tolerant and halophilic GH12 β-1,4-endoglucanase from Trichoderma asperellum ND-1 and its synergistic hydrolysis of lignocellulosic biomass.Int J Biol Macromol. 2024 Jan;254(Pt 1):127650. doi: 10.1016/j.ijbiomac.2023.127650. Epub 2023 Oct 30. Int J Biol Macromol. 2024. PMID: 38287580
-
Cloning and biochemical characterization of an endo-1,4-β-mannanase from the coffee berry borer Hypothenemus hampei.BMC Res Notes. 2013 Aug 22;6:333. doi: 10.1186/1756-0500-6-333. BMC Res Notes. 2013. PMID: 23965285 Free PMC article.
-
Engineering Ligninolytic Consortium for Bioconversion of Lignocelluloses to Ethanol and Chemicals.Protein Pept Lett. 2018;25(2):108-119. doi: 10.2174/0929866525666180122105835. Protein Pept Lett. 2018. PMID: 29359652 Review.
-
Engineering grass biomass for sustainable and enhanced bioethanol production.Planta. 2019 Aug;250(2):395-412. doi: 10.1007/s00425-019-03218-y. Epub 2019 Jun 24. Planta. 2019. PMID: 31236698 Review.
Cited by
-
Research and application progress of microbial β-mannanases: a mini-review.World J Microbiol Biotechnol. 2024 Apr 17;40(6):169. doi: 10.1007/s11274-024-03985-1. World J Microbiol Biotechnol. 2024. PMID: 38630389 Review.
-
Expression of mannanase and glucanases in lettuce chloroplasts and functional evaluation of enzyme cocktail against Candida albicans in oral cancer patient samples.Plant Biotechnol J. 2025 Jul;23(7):2689-2703. doi: 10.1111/pbi.70046. Epub 2025 Apr 18. Plant Biotechnol J. 2025. PMID: 40251764 Free PMC article.
References
-
- Basit A., Liu J., Miao T., Zheng F., Rahim K., Lou H., et al. (2018). Characterization of two endo-β-1,4-xylanases from Myceliophthora thermophila and their saccharifification efficiencies, synergistic with commercial cellulase. Front. Microbiol. 9, 233. 10.3389/fmicb.2018.00233 - DOI - PMC - PubMed
-
- Do B. C., Dang T. T., Berrin J. G., Haltrich D., To K. A., Sigoillot J. C., et al. (2009). Cloning, expression in Pichia pastoris, and characterization of a thermostable GH5 mannan endo-1,4-β-mannosidase from Aspergillus niger BK01. Microb. Cell Fact. 8, 59. 10.1186/1475-2859-8-59 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

