Daily injection of the β2 adrenergic agonist clenbuterol improved poor muscle growth and body composition in lambs following heat stress-induced intrauterine growth restriction
- PMID: 37745251
- PMCID: PMC10516562
- DOI: 10.3389/fphys.2023.1252508
Daily injection of the β2 adrenergic agonist clenbuterol improved poor muscle growth and body composition in lambs following heat stress-induced intrauterine growth restriction
Abstract
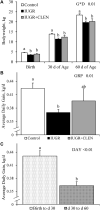
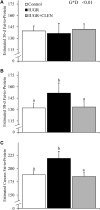
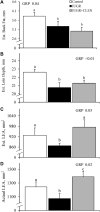
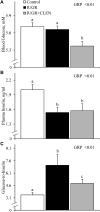
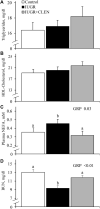
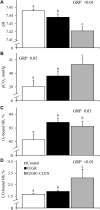
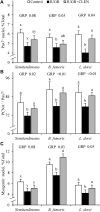
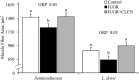
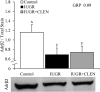
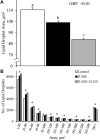
Background: Intrauterine growth restriction (IUGR) is associated with reduced β2 adrenergic sensitivity, which contributes to poor postnatal muscle growth. The objective of this study was to determine if stimulating β2 adrenergic activity postnatal would rescue deficits in muscle growth, body composition, and indicators of metabolic homeostasis in IUGR offspring. Methods: Time-mated ewes were housed at 40°C from day 40 to 95 of gestation to produce IUGR lambs. From birth, IUGR lambs received daily IM injections of 0.8 μg/kg clenbuterol HCl (IUGR+CLEN; n = 11) or saline placebo (IUGR; n = 12). Placebo-injected controls (n = 13) were born to pair-fed thermoneutral ewes. Biometrics were assessed weekly and body composition was estimated by ultrasound and bioelectrical impedance analysis (BIA). Lambs were necropsied at 60 days of age. Results: Bodyweights were lighter (p ≤ 0.05) for IUGR and IUGR+CLEN lambs than for controls at birth, day 30, and day 60. Average daily gain was less (p ≤ 0.05) for IUGR lambs than controls and was intermediate for IUGR+CLEN lambs. At day 58, BIA-estimated whole-body fat-free mass and ultrasound-estimated loin eye area were less (p ≤ 0.05) for IUGR but not IUGR+CLEN lambs than for controls. At necropsy, loin eye area and flexor digitorum superficialis muscles were smaller (p ≤ 0.05) for IUGR but not IUGR+CLEN lambs than for controls. Longissimus dorsi protein content was less (p ≤ 0.05) and fat-to-protein ratio was greater (p ≤ 0.05) for IUGR but not IUGR+CLEN lambs than for controls. Semitendinosus from IUGR lambs had less (p ≤ 0.05) β2 adrenoreceptor content, fewer (p ≤ 0.05) proliferating myoblasts, tended to have fewer (p = 0.08) differentiated myoblasts, and had smaller (p ≤ 0.05) muscle fibers than controls. Proliferating myoblasts and fiber size were recovered (p ≤ 0.05) in IUGR+CLEN lambs compared to IUGR lambs, but β2 adrenoreceptor content and differentiated myoblasts were not recovered. Semitendinosus lipid droplets were smaller (p ≤ 0.05) in size for IUGR lambs than for controls and were further reduced (p ≤ 0.05) in size for IUGR+CLEN lambs. Conclusion: These findings show that clenbuterol improved IUGR deficits in muscle growth and some metabolic parameters even without recovering the deficit in β2 adrenoreceptor content. We conclude that IUGR muscle remained responsive to β2 adrenergic stimulation postnatal, which may be a strategic target for improving muscle growth and body composition in IUGR-born offspring.
Keywords: DOHaD; developmental origins of health and disease; fetal growth restriction; fetal programming; growth efficiency; low birthweight.
Copyright © 2023 Gibbs, Swanson, Beard, Hicks, Most, Beer, Grijalva, Clement, Marks-Nelson, Schmidt, Petersen and Yates.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Anthony W. N., Henry H. L. (2015). Hormones 3ed. Elsvier: Academic Press.
-
- Beermann D. (2002). Beta-adrenergic receptor agonist modulation of skeletal muscle growth. J. Anim. Sci. 80, E18–E23. 10.2527/0021881200800ES10004x - DOI
LinkOut - more resources
Full Text Sources

