Identification and immunogenic potential of glycosylphosphatidylinositol-anchored proteins in Paracoccidioides brasiliensis
- PMID: 37746134
- PMCID: PMC10512324
- DOI: 10.3389/ffunb.2023.1243475
Identification and immunogenic potential of glycosylphosphatidylinositol-anchored proteins in Paracoccidioides brasiliensis
Abstract
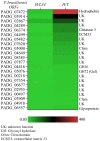
In fungal pathogens the cell wall plays an important role in host-pathogen interactions because its molecular components (e.g., polysaccharides and proteins) may trigger immune responses during infection. GPI-anchored proteins represent the main protein class in the fungal cell wall where they can perform several functions, such as cell wall remodeling and adhesion to host tissues. Genomic analysis has identified the complement of GPI-anchored proteins in many fungal pathogens, but the function has remained unknown for most of them. Here, we conducted an RNA expression analysis of GPI-anchored proteins of Paracoccidioides brasiliensis which causes paracoccidioidomycosis (PCM), an important human systemic mycosis endemic in Latin America. The expression of the GPI-anchored proteins was analyzed by quantitative PCR in both the mycelium and yeast forms. qPCR analysis revealed that the transcript levels of 22 of them were increased in hyphae and 10 in yeasts, respectively, while 14 did not show any significant difference in either form. Furthermore, we cloned 46 open reading frames and purified their corresponding GPI-anchored proteins in the budding yeast. Immunoblot and ELISA analysis of four purified GPI-anchored proteins revealed immune reactivity of these proteins against sera obtained from PCM patients. The information obtained in this study provides valuable information about the expression of many GPI-anchored proteins of unknown function. In addition, based on our immune analysis, some GPI-anchored proteins are expressed during infection and therefore, they might serve as good candidates for the development of new diagnostic methods.
Keywords: GPI-anchored proteins; PCM; Paracoccidioides brasiliensis; Western blot; cell wall.
Copyright © 2023 Gonçales, Portis, dos Reis, Basso Júnior, Martinez, Zhu, Pereira, Soares and Coelho.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Characterization of PbPga1, an antigenic GPI-protein in the pathogenic fungus Paracoccidioides brasiliensis.PLoS One. 2012;7(9):e44792. doi: 10.1371/journal.pone.0044792. Epub 2012 Sep 14. PLoS One. 2012. PMID: 23024763 Free PMC article.
-
In silico identification of glycosylphosphatidylinositol-anchored proteins in Paracoccidioides spp.Future Microbiol. 2021 May;16:589-606. doi: 10.2217/fmb-2020-0282. Epub 2021 May 17. Future Microbiol. 2021. PMID: 33998266
-
rPbPga1 from Paracoccidioides brasiliensis Activates Mast Cells and Macrophages via NFkB.PLoS Negl Trop Dis. 2015 Aug 28;9(8):e0004032. doi: 10.1371/journal.pntd.0004032. eCollection 2015. PLoS Negl Trop Dis. 2015. PMID: 26317855 Free PMC article.
-
Paracoccidioides-host Interaction: An Overview on Recent Advances in the Paracoccidioidomycosis.Front Microbiol. 2015 Nov 25;6:1319. doi: 10.3389/fmicb.2015.01319. eCollection 2015. Front Microbiol. 2015. PMID: 26635779 Free PMC article. Review.
-
The Cell Wall-Associated Proteins in the Dimorphic Pathogenic Species of Paracoccidioides.Curr Protein Pept Sci. 2017;18(11):1074-1089. doi: 10.2174/1389203717666160812233437. Curr Protein Pept Sci. 2017. PMID: 27526924 Review.
References
-
- Bastos K. P., Bailão A. M., Borges C. L., Faria F. P., Felipe M. S., Silva M. G., et al. . (2007). The transcriptome analysis of early morphogenesis in Paracoccidioides brasiliensis mycelium reveals novel and induced genes potentially associated to the dimorphic process. BMC Microbiol. 7, 29. doi: 10.1186/1471-2180-7-29 - DOI - PMC - PubMed
-
- Castro Nda S., de Castro K. P., Orlandi I., Feitosa Ldos S., Rosa e Silva L. K., Vainstein M. H., et al. . (2009). Characterization and functional analysis of the beta-1,3-glucanosyltransferase 3 of the human pathogenic fungus Paracoccidioides brasiliensis. FEMS Yeast Res. 9 (1), 103–114. doi: 10.1111/j.1567-1364.2008.00463.x - DOI - PubMed
LinkOut - more resources
Full Text Sources

