Correlating physicochemical and biological properties to define critical quality attributes of a rAAV vaccine candidate
- PMID: 37746246
- PMCID: PMC10512015
- DOI: 10.1016/j.omtm.2023.06.004
Correlating physicochemical and biological properties to define critical quality attributes of a rAAV vaccine candidate
Abstract
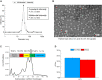
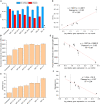
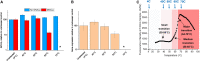
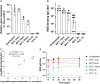
Recombinant adeno-associated viruses (rAAVs) are a preferred vector system in clinical gene transfer. A fundamental challenge to formulate and deliver rAAVs as stable and efficacious vaccines is to elucidate interrelationships between the vector's physicochemical properties and biological potency. To this end, we evaluated an rAAV-based coronavirus disease 2019 (COVID-19) vaccine candidate that encodes the Spike antigen (AC3) and is produced by a commercially viable process. First, state-of-the-art analytical techniques were employed to determine key structural attributes of AC3, including primary and higher-order structures, particle size, empty/full capsid ratios, aggregates, and multi-step thermal degradation pathway analysis. Next, several quantitative potency measures for AC3 were implemented, and data were correlated with the physicochemical analyses on thermally stressed and control samples. Results demonstrate links between decreasing AC3 physical stability profiles, in vitro transduction efficiency in a cell-based assay, and, importantly, in vivo immunogenicity in a mouse model. These findings are discussed in the general context of future development of rAAV-based vaccine candidates as well as specifically for the rAAV vaccine application under study.
Keywords: COVID-19; adeno-associated virus; analytical characterization; biological properties; formulation; immunogenicity; physicochemical properties; stability; transduction efficiency; vaccine.
© 2023 The Author(s).
Conflict of interest statement
L.H.V. is an inventor on AAVCOVID and AAV vaccine patent applications and a founder and employee of ciendias bio, a vaccine biotechnology company. N.Z. is an inventor on AAVCOVID and AAV vaccine patent applications.
Figures





References
-
- WHO Timeline: WHO's COVID-19 response. 2022. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/interact...
-
- U.S. GAO. Operation Warp Speed: Accelerated COVID-19 Vaccine Development Status and Efforts to Address Manufacturing Challenges. 2021. https://www.gao.gov/products/gao-21-319
-
- Nasreen S., Chung H., He S., Brown K.A., Gubbay J.B., Buchan S.A., Fell D.B., Austin P.C., Schwartz K.L., Sundaram M.E., et al. Canadian Immunization Research Network CIRN Provincial Collaborative Network PCN Investigators Effectiveness of COVID-19 vaccines against symptomatic SARS-CoV-2 infection and severe outcomes with variants of concern in Ontario. Nat. Microbiol. 2022;7:379–385. doi: 10.1038/s41564-021-01053-0. - DOI - PubMed
LinkOut - more resources
Full Text Sources

