Cyclophosphamide augments the efficacy of in situ vaccination in a mouse melanoma model
- PMID: 37746303
- PMCID: PMC10516537
- DOI: 10.3389/fonc.2023.1200436
Cyclophosphamide augments the efficacy of in situ vaccination in a mouse melanoma model
Abstract
Introduction: We have previously shown that an intratumoral (IT) injection of the hu14.18-IL2 immunocytokine (IC), an anti-GD2 antibody linked to interleukin 2, can serve as an in situ vaccine and synergize with local radiotherapy (RT) to induce T cell-mediated antitumor effects. We hypothesized that cyclophosphamide (CY), a chemotherapeutic agent capable of depleting T regulatory cells (Tregs), would augment in situ vaccination. GD2+ B78 mouse melanoma cells were injected intradermally in syngeneic C57BL/6 mice.
Methods: Treatments with RT (12Gy) and/or CY (100 mg/kg i.p.) started when tumors reached 100-300 mm3 (day 0 of treatment), followed by five daily injections of IT-IC (25 mcg) on days 5-9. Tumor growth and survival were followed. In addition, tumors were analyzed by flow cytometry.
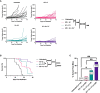
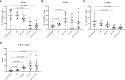
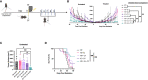
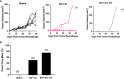
Results: Similar to RT, CY enhanced the antitumor effect of IC. The strongest antitumor effect was achieved when CY, RT and IC were combined, as compared to combinations of IC+RT or IC+CY. Flow cytometric analyses showed that the combined treatment with CY, RT and IC decreased Tregs and increased the ratio of CD8+ cells/Tregs within the tumors. Moreover, in mice bearing two separate tumors, the combination of RT and IT-IC delivered to one tumor, together with systemic CY, led to a systemic antitumor effect detected as shrinkage of the tumor not treated directly with RT and IT-IC. Cured mice developed immunological memory as they were able to reject B78 tumor rechallenge.
Conclusion: Taken together, these preclinical results show that CY can augment the antitumor efficacy of IT- IC, given alone or in combination with local RT, suggesting potential benefit in clinical testing of these combinations.
Keywords: cyclophosphamide; immunocytokine; in situ vaccine.; melanoma; radiation.
Copyright © 2023 Tsarovsky, Felder, Heck, Slowinski, Rasmussen, VandenHeuvel, Zaborek, Morris, Erbe, Sondel and Rakhmilevich.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Radiation to all macroscopic sites of tumor permits greater systemic antitumor response to in situ vaccination.J Immunother Cancer. 2023 Jan;11(1):e005463. doi: 10.1136/jitc-2022-005463. J Immunother Cancer. 2023. PMID: 36639155 Free PMC article.
-
Short-course neoadjuvant in situ vaccination for murine melanoma.J Immunother Cancer. 2022 Jan;10(1):e003586. doi: 10.1136/jitc-2021-003586. J Immunother Cancer. 2022. PMID: 35039460 Free PMC article.
-
Intratumoral injection reduces toxicity and antibody-mediated neutralization of immunocytokine in a mouse melanoma model.J Immunother Cancer. 2020 Oct;8(2):e001262. doi: 10.1136/jitc-2020-001262. J Immunother Cancer. 2020. PMID: 33115944 Free PMC article.
-
Tumor-Specific Inhibition of In Situ Vaccination by Distant Untreated Tumor Sites.Cancer Immunol Res. 2018 Jul;6(7):825-834. doi: 10.1158/2326-6066.CIR-17-0353. Epub 2018 May 10. Cancer Immunol Res. 2018. PMID: 29748391 Free PMC article.
-
In situ Vaccine Plus Checkpoint Blockade Induces Memory Humoral Response.Front Immunol. 2020 Jul 24;11:1610. doi: 10.3389/fimmu.2020.01610. eCollection 2020. Front Immunol. 2020. PMID: 32849544 Free PMC article.
References
-
- Salas-Benito D, Pérez-Gracia JL, Ponz-Sarvisé M, Rodriguez-Ruiz ME, Martínez-Forero I, Castañón E, et al. . Paradigms on immunotherapy combinations with chemotherapy. Cancer Discov (2021) 11(6):1353–67. doi: 10.1158/2159-8290.CD-20-1312 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

