Zinc nanoparticles ameliorated obesity-induced cardiovascular disease: role of metabolic syndrome and iron overload
- PMID: 37749096
- PMCID: PMC10519991
- DOI: 10.1038/s41598-023-42550-y
Zinc nanoparticles ameliorated obesity-induced cardiovascular disease: role of metabolic syndrome and iron overload
Erratum in
-
Author Correction: Zinc nanoparticles ameliorated obesity-induced cardiovascular disease: role of metabolic syndrome and iron overload.Sci Rep. 2024 Feb 14;14(1):3746. doi: 10.1038/s41598-024-53947-8. Sci Rep. 2024. PMID: 38355658 Free PMC article. No abstract available.
Abstract
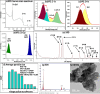
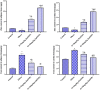
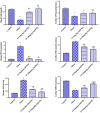
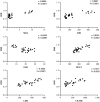
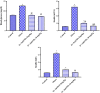
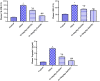
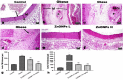
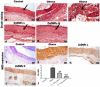
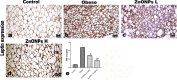
Obesity is a complicated disease characterized by abundant fat accumulation. It is associated with cardiovascular disease. The current study aimed to appreciate the role of synthesized zinc oxide nanoparticles (ZnONPs) (18.72 nm in size) in curbing cardiovascular disease in an obesity model of a high fat/sucrose diet in male rats. For 16 weeks, 24 rats were fed a high-fat diet and a 25% sucrose solution to develop obesity, and after that, the rats were randomly allocated into four groups of rats. Group 1 served as the control group and consisted of normal, non-obese rats. Group 2 comprised obese rats that were injected with an equivalent volume of a neutral substance, serving as vehicle control. In Group 3 or 4, obese rats were treated with an intraperitoneal injection of 5 or 10mg/kg of zinc oxide nanoparticles (ZnONPs) for eight weeks. The treatment of obese rats with ZnONPs decreased plasma levels of monocyte chemoattractant Protein-1 (MCP-1), resistin, ENA78, tumor necrosis factor-alpha (TNF-α), interleukin 6 (IL6), and C reactive protein (CRP). Also, the remediation of obese rats with ZnONPs led to a significant decrease in body mass index (BMI), body weight gain, leptin, cholesterol, triglycerides, LDL (Low-density lipoprotein), glucose, and insulin resistance index (HOMA-IR). Moreover, ZnONPs treatment lowered troponin, creatine phosphokinase-MB (CK-MB), lactate dehydrogenase (LDH), cardiac or adipose tissue iron content, and malondialdehyde (MDA) either in blood or heart tissue. Otherwise, treating obese rats with ZnONPs enhanced plasma adiponectin levels, cardiac-reduced glutathione (GSH), and superoxide dismutase (SOD). In addition, ZnONPs displayed a significant influence on the cardiovascular system since they combat the rise in blood pressure and the pathological changes of the heart and aorta besides maintaining plasma nitric oxide levels. The results showed a positive correlation between BMI and MDA, MPC-1, CK-MB, and LDH. ZnONPs are convenient in treating cardiovascular disease in obese rats via reduced blood pressure, oxidative stress, cardiac iron accumulation, insulin resistance, and inflammatory markers.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Abdelmottaleb SAM, et al. Goldenberry (Physalis peruviana) alleviates hepatic oxidative stress and metabolic syndrome in obese rats. J. Appl. Pharm. Sci. 2022 doi: 10.7324/japs.2022.121115. - DOI
-
- Sheibani H, et al. A comparison of body mass index and body fat percentage for predicting cardiovascular disease risk. Transl. Metab. Syndr. Res. 2020;3:29–34.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

