Allometrically scaling tissue forces drive pathological foreign-body responses to implants via Rac2-activated myeloid cells
- PMID: 37749310
- PMCID: PMC10651488
- DOI: 10.1038/s41551-023-01091-5
Allometrically scaling tissue forces drive pathological foreign-body responses to implants via Rac2-activated myeloid cells
Abstract
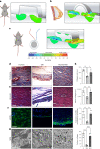
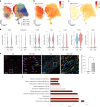
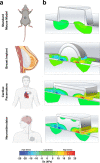
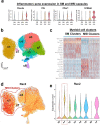
Small animals do not replicate the severity of the human foreign-body response (FBR) to implants. Here we show that the FBR can be driven by forces generated at the implant surface that, owing to allometric scaling, increase exponentially with body size. We found that the human FBR is mediated by immune-cell-specific RAC2 mechanotransduction signalling, independently of the chemistry and mechanical properties of the implant, and that a pathological FBR that is human-like at the molecular, cellular and tissue levels can be induced in mice via the application of human-tissue-scale forces through a vibrating silicone implant. FBRs to such elevated extrinsic forces in the mice were also mediated by the activation of Rac2 signalling in a subpopulation of mechanoresponsive myeloid cells, which could be substantially reduced via the pharmacological or genetic inhibition of Rac2. Our findings provide an explanation for the stark differences in FBRs observed in small animals and humans, and have implications for the design and safety of implantable devices.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures









References
-
- The Global Market for Medical Devices, 10th edn (Kalorama Information, 2019).
-
- Dolan, E. B. et al. An actuatable soft reservoir modulates host foreign body response. Sci. Robot.10.1126/scirobotics.aax7043 (2019). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

