Assessing the safety and activity of cabozantinib combined with lanreotide in gastroenteropancreatic and thoracic neuroendocrine tumors: rationale and protocol of the phase II LOLA trial
- PMID: 37752423
- PMCID: PMC10523723
- DOI: 10.1186/s12885-023-11287-2
Assessing the safety and activity of cabozantinib combined with lanreotide in gastroenteropancreatic and thoracic neuroendocrine tumors: rationale and protocol of the phase II LOLA trial
Abstract
Background: Well-differentiated (WD) neuroendocrine tumors (NETs) are a group of rare neoplasms with limited therapeutic options. Cabozantinib is an inhibitor of multiple tyrosine kinases with a pivotal role in NET pathogenesis, including c-MET and Vascular Endothelial Growth Factor Receptor 2 (VEGFR2). LOLA is the first prospective phase II trial aiming to assess the safety and activity of cabozantinib combined with lanreotide in WD NETs of gastroenteropancreatic (GEP), thoracic and of unknown origin.
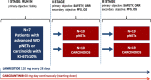
Methods: This is a multicenter, open-label, double-cohort, non comparative, non-randomized, three-stage phase II trial. Eligible patients have to meet the following inclusion criteria: diagnosis of advanced or metastatic, progressive, non-functioning WD thoracic NETs, GEP-NETs or NETs of unknown origin with Ki67 ≥ 10%; positive 68 Ga-PET uptake or somatostatin receptor 2 immunohistochemical (IHC) stain; maximum 1 prior systemic regimen for metastatic disease. Two cohorts will be considered: pNETs and carcinoids (typical or atypical lung and thymus NETs, gastro-intestinal NETs or NETs of unknown origin). In stage I, the primary objective is to find the optimal dose of cabozantinib in combination with lanreotide and to evaluate the safety of the combination (percentage of patients experiencing grade 3-5 toxicities according to NCI-CTCAE version 5.0). Starting dose of cabozantinib is 60 mg/day continuously, plus lanreotide 120 mg every 28 days. In stage II and III, co-primary endpoints are safety and overall response rate (ORR) according to RECIST version 1.1. The uninteresting antitumor activity is fixed in ORR ≤ 5%. Secondary endpoints are progression-free survival and overall survival. Exploratory objectives include the assessment of c-MET, AXL and VEGFR2 IHC expression, to identify predictive or prognostic tissue biomarkers. Enrolment started in July 2020, with an expected trial duration of 42 months comprehensive of accrual, treatment and follow-up. Considering a drop-out rate of 5%, the maximum number of enrolled patients will be 69.
Discussion: Supported by a solid rationale, the trial has the potential to generate milestone data about the synergistic effects of cabozantinib plus lanreotide in a group of NET patients with relatively aggressive disease and limited therapeutic options.
Trial registration: LOLA is registered at ClinicalTrials.gov (NCT04427787) and EudraCT (2019-004506-10).
Keywords: Cabozantinib; Clinical trial; Lanreotide; Neuroendocrine tumors; Somatostatin analogs; Tyrosine kinase inhibitors.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
Dr. Pusceddu reported receiving honoraria from Novartis, Ipsen, Pfizer, Merck Serono, and Advanced Accelerator Applications (AAA); receiving grants from Ipsen and Pfizer; and receiving personal fees from AAA, Novartis, and Merck outside the submitted work. Dr. Prinzi reported receiving honoraria and travel accommodations from Novartis, Ipsen, Pfizer, Merck Serono, and Italfarmaco and receiving personal fees from Merck outside the submitted work. Dr. Panzuto reported receiving honoraria for speaker and advisory roles from Novartis, for an advisory role from Ipsen, and for consulting from AAA and receiving travel accommodations from Pfizer. Dr. Corti reported receiving nonfinancial support from AAA and Ipsen outside the submitted work. Dr. Porcu reported receiving honoraria for consulting from Ipsen and Italfarmaco. Dr. de Braud reported receiving honoraria for serving on advisory boards or speakers bureaus for Amgen, Novartis, Roche, Incyte, EMD Serono, Bristol Myers Squibb, Roche, Pfizer, Menarini, Sanofi, Healthcare Research & Pharmacoepidemiology, and Dephaforum; receiving research funding from Novartis, Roche, Merck Sharp & Dohme, Merck Serono, Pfizer, Servier, and Nerviano Medical Sciences; and receiving honoraria and/or personal fees from Roche, EMD Serono, Nerviano Medical Sciences, Sanofi, MSD, Novartis, Incyte, Bristol Myers Squibb, Menarini, Merck, Pfizer, Healthcare Research & Pharmacoloepidemiology, Servier, Amgen, and Dephaforum outside the submitted work. No other disclosures were reported.
Figures

References
-
- Rinke A, Muller H, Schade-Brittinger C, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: A report from the PROMID study group. J Clin Oncol. 2009;27(28):4656–4663. doi: 10.1200/JCO.2009.22.8510. - DOI - PubMed
-
- Caplin ME, Pavel M, Cwikla JB, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371(16):1556–1557. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

