Endogenous opioid system modulates conditioned cocaine reward in a sex-dependent manner
- PMID: 37753570
- PMCID: PMC11974355
- DOI: 10.1111/adb.13328
Endogenous opioid system modulates conditioned cocaine reward in a sex-dependent manner
Abstract
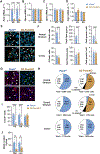
Cocaine predictive cues and contexts exert powerful control over behaviour and can incite cocaine seeking and taking. This type of conditioned behaviour is encoded within striatal circuits, and these circuits and behaviours are, in part, regulated by opioid peptides and receptors expressed in striatal medium spiny neurons. We previously showed that augmenting levels of the opioid peptide enkephalin in the striatum facilitates acquisition of cocaine conditioned place preference (CPP), while opioid receptor antagonists attenuate expression of cocaine CPP. However, whether striatal enkephalin is necessary for acquisition of cocaine CPP and maintenance during extinction remains unknown. To address this, we generated mice with a targeted deletion of enkephalin from dopamine D2-receptor expressing medium spiny neurons and tested them in a cocaine CPP paradigm. Low striatal enkephalin levels did not attenuate acquisition of CPP. However, expression of preference, assessed after acute administration of the opioid receptor antagonist naloxone, was blocked in females, regardless of genotype. When saline was paired with the cocaine context during extinction sessions, females, regardless of genotype, extinguished preference faster than males, and this was prevented by naloxone when paired with the cocaine context. We conclude that while striatal enkephalin is not necessary for acquisition, expression, or extinction of cocaine CPP, expression and extinction of cocaine preference in females is mediated by an opioid peptide other than striatal enkephalin. The unique sensitivity of females to opioid antagonists suggests sex should be a consideration when using these compounds in the treatment of cocaine use disorder.
Keywords: cocaine reward; enkephalin; striatum.
© 2023 The Authors. Addiction Biology published by John Wiley & Sons Ltd on behalf of Society for the Study of Addiction.
Conflict of interest statement
Competing Interests
We have no competing interests to report.
Figures
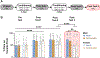


Similar articles
-
Mu opioid receptors expressed in striatal D2 medium spiny neurons have divergent contributions to cocaine and morphine reward.Neuroscience. 2025 Mar 5;568:273-284. doi: 10.1016/j.neuroscience.2025.01.034. Epub 2025 Jan 18. Neuroscience. 2025. PMID: 39832666
-
Striatal enkephalin supports maintenance of conditioned cocaine reward during extinction.bioRxiv [Preprint]. 2023 Feb 24:2023.02.23.529807. doi: 10.1101/2023.02.23.529807. bioRxiv. 2023. PMID: 36865224 Free PMC article. Preprint.
-
Dopamine D2 receptors bidirectionally regulate striatal enkephalin expression: Implications for cocaine reward.Cell Rep. 2022 Sep 27;40(13):111440. doi: 10.1016/j.celrep.2022.111440. Cell Rep. 2022. PMID: 36170833 Free PMC article.
-
Naloxone effects on extinction of ethanol- and cocaine-induced conditioned place preference in mice.Psychopharmacology (Berl). 2017 Sep;234(18):2747-2759. doi: 10.1007/s00213-017-4672-z. Epub 2017 Jun 26. Psychopharmacology (Berl). 2017. PMID: 28653079 Free PMC article.
-
Endogenous opiates and behavior: 2012.Peptides. 2013 Dec;50:55-95. doi: 10.1016/j.peptides.2013.10.001. Epub 2013 Oct 12. Peptides. 2013. PMID: 24126281 Review.
Cited by
-
Dopamine Pharmacodynamics: New Insights.Int J Mol Sci. 2024 May 13;25(10):5293. doi: 10.3390/ijms25105293. Int J Mol Sci. 2024. PMID: 38791331 Free PMC article. Review.
-
Mu opioid receptors expressed in striatal D2 medium spiny neurons have divergent contributions to cocaine and morphine reward.Neuroscience. 2025 Mar 5;568:273-284. doi: 10.1016/j.neuroscience.2025.01.034. Epub 2025 Jan 18. Neuroscience. 2025. PMID: 39832666
-
Shifted Balance Between Ventral Striatal Prodynorphin and Proenkephalin Biases Development of Cocaine Place Avoidance.Addict Biol. 2025 Jul;30(7):e70055. doi: 10.1111/adb.70055. Addict Biol. 2025. PMID: 40619251 Free PMC article.
References
-
- Gutiérrez-Cuesta J, Burokas A, Mancino S, Kummer S, Martín-García E, Maldonado R. Effects of genetic deletion of endogenous opioid system components on the reinstatement of cocaine-seeking behavior in mice. Neuropsychopharmacology. 2014;39(13):2974–2988. doi:10.1038/npp.2014.149 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

