NMOSD IgG Impact Retinal Cells in Murine Retinal Explants
- PMID: 37754247
- PMCID: PMC10529972
- DOI: 10.3390/cimb45090463
NMOSD IgG Impact Retinal Cells in Murine Retinal Explants
Abstract
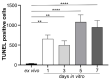
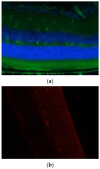
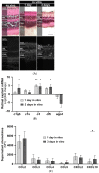
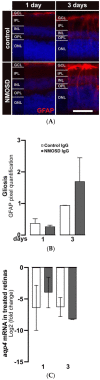
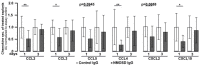
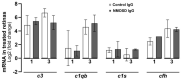
Neuromyelitis optica spectrum disorders (NMOSD) are chronic inflammatory diseases of the central nervous system, characterized by autoantibodies against aquaporin-4. The symptoms primarily involve severe optic neuritis and longitudinally extensive transverse myelitis. Although the disease progression is typically relapse-dependent, recent studies revealed retinal neuroaxonal degeneration unrelated to relapse activity, potentially due to anti-aquaporin-4-positive antibodies interacting with retinal glial cells such as Müller cells. In this exploratory study, we analysed the response of mouse retinal explants to NMOSD immunoglobulins (IgG). Mouse retinal explants were treated with purified IgG from patient or control sera for one and three days. We characterized tissue response patterns through morphological changes, chemokine secretion, and complement expression. Mouse retinal explants exhibited a basic proinflammatory response ex vivo, modified by IgG addition. NMOSD IgG, unlike control IgG, increased gliosis and decreased chemokine release (CCL2, CCL3, CCL4, and CXCL-10). Complement component expression by retinal cells remained unaltered by either IgG fraction. We conclude that human NMOSD IgG can possibly bind in the mouse retina, altering the local cellular environment. This intraretinal stress may contribute to retinal degeneration independent of relapse activity in NMOSD, suggesting a primary retinopathy.
Keywords: Müller cell; NMOSD; autoantibodies; chemokine; complement; local; mouse retinal explants; retina.
Conflict of interest statement
T.K. has received speaker honoraria and/or personal fees for advisory boards from Novartis Pharma, Roche Pharma, Alexion/Astra Zeneca, and Biogen; the institution she works for has received grant support for her research from Bayer-Schering A.G., Novartis, and Chugai Pharma. J.H. reports grants from Friedrich-Baur-Stiftung, Merck, and Horizon; personal fees and non-financial support from Alexion, Horizon, Roche, Merck, Novartis, Biogen, B.M.S., and Janssen; and non-financial support from the Guthy-Jackson Charitable Foundation and The Sumaira Foundation. The other authors declare no conflict of interest.
Figures


References
-
- Filippatou A.G., Vasileiou E.S., He Y., Fitzgerald K.C., Kalaitzidis G., Lambe J., Mealy M.A., Levy M., Liu Y., Prince J.L., et al. Evidence of subclinical quantitative retinal layer abnormalities in AQP4-IgG seropositive NMOSD. Mult. Scler. 2021;27:1738–1748. doi: 10.1177/1352458520977771. - DOI - PMC - PubMed
-
- Motamedi S., Oertel F.C., Yadav S.K., Kadas E.M., Weise M., Havla J., Ringelstein M., Aktas O., Albrecht P., Ruprecht K., et al. Altered fovea in AQP4-IgG-seropositive neuromyelitis optica spectrum disorders. Neurol. Neuroimmunol. Neuroinflammation. 2020;7:e805. doi: 10.1212/NXI.0000000000000805. - DOI - PMC - PubMed
-
- Oertel F.C., Havla J., Roca-Fernández A., Lizak N., Zimmermann H., Motamedi S., Borisow N., White O.B., Bellmann-Strobl J., Albrecht P., et al. Retinal ganglion cell loss in neuromyelitis optica: A longitudinal study. J. Neurol. Neurosurg. Psychiatry. 2018;89:1259–1265. doi: 10.1136/jnnp-2018-318382. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

