Efflux pump inhibitory potential of indole derivatives as an arsenal against norA over-expressing Staphylococcus aureus
- PMID: 37754560
- PMCID: PMC10581058
- DOI: 10.1128/spectrum.04876-22
Efflux pump inhibitory potential of indole derivatives as an arsenal against norA over-expressing Staphylococcus aureus
Abstract
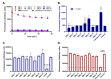
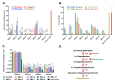
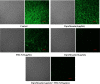
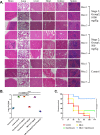
NorA, an extensively studied efflux pump in Staphylococcus aureus, has been connected to fluoroquinolone, antiseptic, and disinfection resistance. Several studies have also emphasized how efflux pumps, including NorA, function as the first line of defense of S. aureus against antibiotics. In this study, we have screened some chemically synthesized indole derivatives for their activity as efflux pump inhibitors (EPIs). The derivative SMJ-5 was found to be a potent NorA efflux pump inhibitor among the screened indole derivatives, owing to increased ethidium bromide and norfloxacin accumulation in norA over-expressing S. aureus. The combination of SMJ-5 and ciprofloxacin demonstrated the eradication of S. aureus biofilm and prolonged the post-antibiotic effect more than ciprofloxacin alone. SMJ-5 was able to inhibit staphyloxanthin virulence. In in vitro time-kill trials and in vivo efficacy investigations, the combination enhanced the bactericidal activity of ciprofloxacin against S. aureus. Additionally, reverse transcription PCR results revealed that SMJ-5 also inhibits the NorA efflux pump indirectly at the transcriptional level. IMPORTANCE The NorA efflux pump is the most effective resistance mechanism in S. aureus. The clinical importance of NorA efflux pumps is demonstrated by the expression of pump genes in S. aureus strains in response to fluoroquinolones and biocides. Along with the repercussions of decreased fluoroquinolone sensitivity, increasing expression of efflux pump genes by their substrate necessitates the importance of efflux pump inhibitors. Reserpine and verapamil are clinically used to treat ailments and have proven NorA inhibitors, but, unfortunately, the concentration needed for these drugs to inhibit the pump is not safe in clinical settings. In the current study, we have screened some indole derivatives, and among them, SMJ-5 was reported to potentiate norfloxacin and ciprofloxacin at their sub-inhibitory concentration by inhibiting the norA gene transcriptionally. Here we highlight the promising points of this study, which could serve as a model to design a therapeutic EPI candidate against norA over-expressing S. aureus.
Keywords: Staphylococcus aureus; efflux pump inhibitors (EPIs); major facilitator superfamily (MFS); multidrug resistance (MDR); norA efflux pump.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




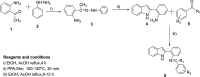
References
-
- Gagliotti C, Balode A, Baquero F, Degener J, Grundmann H, Gür D, Jarlier V, Kahlmeter G, Monen J, Monnet DL, Rossolini GM, Suetens C, Weist K, Heuer O, EARS-Net Participants (Disease Specific Contact Points for AMR) . 2011. Escherichia coli and Staphylococcus aureus: bad news and good news from the European antimicrobial resistance surveillance network (EARS-net, formerly EARSS), 2002 to 2009. Euro Surveill 16:19819. doi:10.2807/ese.16.11.19819-en - DOI - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

